Childhood sarcoidosis is a rare but extremely challenging pathology, for early diagnosis and management, precisely because of the multitude of clinical phenotypic aspects that it has within its progressive multisystemic involvement. Its incidence is only 2.3-11 per 100000 person/year and in children 0.22-0.29 per 100000 person/year, the age at diagnosis is frequently between 13-15 years, although the actual onset may sometimes be in infancy (1).However, only 70% of cases are diagnosed in adults aged 25-40 years, the rest being found in children. (2).Sarcoidosis is categorized as a chronic immune inflammatory disease, which induces infiltration of various tissues by non-caesous granulomas. The central pathogenic element that generates the inflammatory immune cascade is the TH1-TH17 cytokine stimulation induced by dendritic cells and the monocyte-macrophage system through the HLA class 2 system and therefore the basic treatment of these patients is immunosuppressive. Moreover, the genetic component of sarcoidosis is presently demonstrated and involves in addition to mutations of the HLA class 2 complex (HLA-A, -B, -DQB1, -DRB1-3) chromosomal mutations and genetic polymorphisms (BTNL2, ANXAII). Although its evolution is extensive most of the time, with one or more affected organs, only 1/3 of the cases seem to have a progressive, chronic evolution, the rest registering a self-limited evolution over an average variable period of 1-3 years.
The clinical picture at diagnosis is characterized by non-specific manifestations such as recurrent fever, weight loss, asthenia, sweating. Respiratory involvement of mild to moderate dyspnea, occasional chest pain, chronic cough, is the most common. In 50% of cases, progressive pulmonary dysfunction is manifested by a restrictive component as a consequence of early alveolitis progressing to pulmonary fibrosis. In 15% of cases, however, obstructive pulmonary dysfunction may be caused by compression of the airways by intrabronchial sarcoid granuloma, pulmonary or mediastinal lymph nodes or by the development of bronchiectasis. On the other hand, 85% of children with pulmonary sarcoidosis have evolving systemic involvement.(1)
Hepatosplenomegaly affects 43-49% of children with sarcoidosis but clinically significant liver dysfunction is rare. Mild to moderate elevation of transaminase values is common, but severe liver involvement with cirrhosis and portal hypertension or splenic involvement is uncommon in children. Peripheral adenopathy may be objectifiable in 40-70% of cases, mobile , painless and nonsuppurative.(1,3)
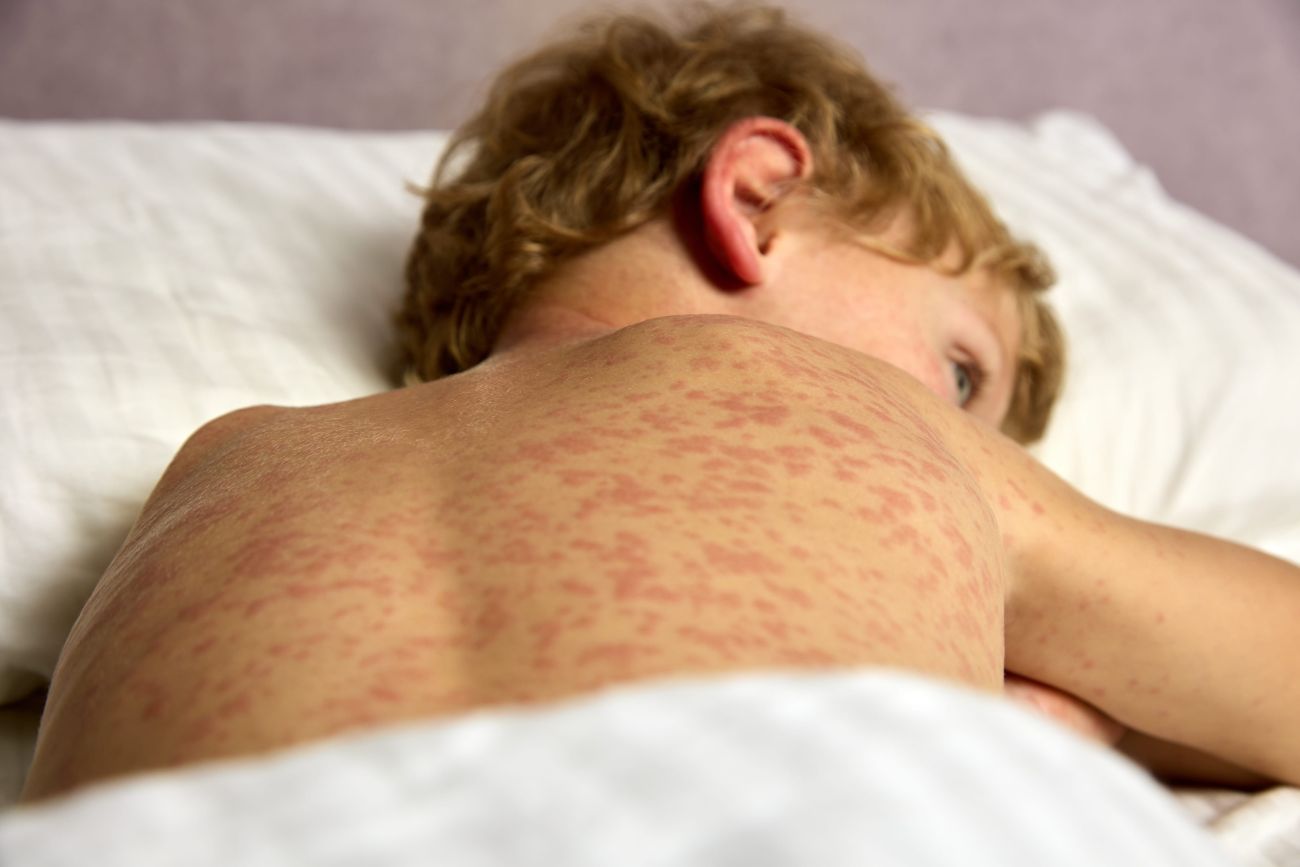
Cutaneous involvement may be present in approximately 24-40% of older children and 77% of young children with sarcoidosis. Sometimes the appearance is of the urticarial type with little itching, sometimes there may be soft, red to yellowish or purplish papules, commonly seen on the face or erythematous macular lesions, ihtiosiform eruptions or erythematous nodosal appearance especially on the limbs which must be differentiated from bacterial infectious (streptococcus, tuberculosis).(1,3)
Uveitis may occur in 39% of cases, but other ocular lesions such as recurrent conjunctivitis and/or iridocyclitis may also occur. Musculoskeletal pain in 15% of cases, sometimes associated with the appearance of active arthritis, may lead to confusion with the onset of juvenile idiopathic arthritis (JIA). Less common is central and/or peripheral neurological involvement, manifested as peripheral neuropathy, migraine headache syndrome, meningitis and also cardiac involvement, accompanied by pathologic murmurs in 5% of cases.(1)
The young child under the age of 5 years may present a particular form of sarcoidosis with early onset, which is a sporadic form, with autosomal dominant transmission, but without familial involvement and which combines for positive diagnosis the classic triad: eczema, uveitis and arthritis. In this form, the eczema-like cutaneous component, sometimes pruritic, lends itself to differential diagnosis with allergologic skin disorders (urticaria, atopic dermatitis), and is frequently included in this category. Ophthalmologic involvement occurs in most cases (58-90%) in the form of acute or chronic uveitis, which can range from isolated iridocyclitis to bilateral panuveitis and which in 16% of cases can lead to blindness. Unlike the uveitis associated with juvenile idiopathic juvenile arthritis, sarcoidosis uveitis is granulomatous and affects the posterior rather than the anterior segment.(4)
The osteo-articular symptomatology of early-onset sarcoidosis, cited with an incidence of 45-58% of cases, frequently presents as arthralgia or less rarely arthritis. In this case the large joints are mainly affected and may sometimes present with joint effusion. At other times we may find the appearance of tenosynovitis, a tenosynovial cyst secondary to granulomatous inflammation. In particular, unlike JIA, sarcoidotic arthritis rarely induces bone and joint lesions or significant motor dysfunction.
Another form of sarcoidosis found in young children, but with autosomal dominant familial transmission, is Blau syndrome. At present, 18 genetic mutations related to the NOD-2 gene have been identified in this subtype of sarcoidosis. (5).
Both forms can develop in 80-100% of cases disabling sequelae in the eye, joints or systemic extension: hepatosplenic (52%), adenopathy (42%), parotid gland (13%), lung (13-22%). More rarely, cardiac (pericarditis, myocarditis, intraventricular thrombosis), renal (vasculitis) and CNS involvement has been reported.
Acute sarcoidosis, manifested as either Heerfordt’s syndrome or Lofgren’s syndrome, is rare in children and is usually seen in adults. The febrile syndrome associated with systemic involvement, but also the self-limited evolution under treatment, with favorable prognosis, is a characteristic feature.(6)
Although currently, there are no absolute diagnostic tests or biomarkers for screening or disease activity, the diagnosis will take into account the age of onset, suggestive clinical picture, but also the histopathologic and genetic examination (NOD 2 positive/no) especially for early onset sarcoidosis under 5 years of age.(9, 10) As a result, the positive diagnosis in childhood sarcoidosis is one of exclusion and is based, in addition to the clinical manifestations listed above and particularized by form and age, on specific laboratory investigations, which include: immunophenotyping for the study of lymphocyte lines and CD4/CD8 ratio in broncho-alveolar lavage fluid; specific histopathologic aspects by biopsy of various tissues (skin, liver, lymph nodes, salivary gland, medulogram); complementary paraclinical imaging examinations of the affected organ (CT/MRI/ EBUS/TBNA) completed with ophthalmologic, cardiologic, neurologic, nephrologic and pneumologic examinations as appropriate. (3,7) Keeping these aspects in mind, pathologies expressed by granulomatous inflammation such as tuberculosis, lymphomas, histoplasmosis, coccidiomycosis, aspergillosis, eosinophilic granulomatosis with polyangiitis, primary immune deficiencies, Crohn’s disease, bacterial and fungal infections should be excluded.(8)
Patients diagnosed with common variable immunodeficiency, which is also associated with systemic sarcoidosis, are in a special category. They were found to be more prone to hepatosplenomegaly, recurrent infections and autoimmune diseases versus those without immunodeficiency.(8) On the other hand, the CD4/CD8 ratio in bronchoalveolar lavage fluid is also lower compared to patients without immune deficiency. Furthermore, the CT lung imaging patterns were also different: peribronchovascular nodules and micronodules were more frequent in sarcoidosis without immunodeficiency. In this category of patients the monitoring of serum immunoglobulin levels is very important and individualizes specific immunosuppressive therapy associated with iv immunoglobulin support.
The basic treatment in sarcoidosis is immunosuppression initiated with systemic corticosteroid therapy, preferably pulsterapia, which is mainly addressed to the extensive and rapidly progressive forms of the disease and especially to those with severe lung involvement and progressive functional deterioration. In the case of non-responsiveness even to high doses of cortisone / corticosteroid resistance or adverse reactions to it, corticodependence, immunosuppressive therapy with methotrexate or azathioprine can be used, and in refractory therapeutic situations we can use anti-cytokine biological agents – anti TNF-alpha, anti-IL1, anti-IL6.(8) Thus studies show that at diagnosis 94.2% of patients receive corticosteroid therapy and 48.1% of patients are switched to another immunosuppressive therapy.(11) Although there are no predictive prognostic markers for the evolution of sarcoidosis in children, however, studies show that patients under 10 years of age at diagnosis who responded favorably to corticosteroid therapy with improvement of pulmonary functional parameters, have a progression to remission and stabilization in 70% of cases vs patients diagnosed over this age.(1) However some complications such as treatment-resistant pulmonary sarcoidosis, cardiac sarcoidosis, neurosarcoidosis and/or multi-organ involvement are associated with a relatively high risk of death . Adult with pediatric-onset sarcoidosis has demonstrated that in 50% of cases the disease is perfectly therapeutically controlled and may not require chronic treatment. However, in the remaining cases, they continue to present predominantly pulmonary (47%), ocular (36.6%) and hepatic (23.3%) involvement, they associate an undulating course with relapses, and 4.8% of them remain with uncontrolled disease and 1.2% end up needing lung transplantation (11). In conclusion, pediatric sarcoidosis should be evoked in the presence of isolated signs of organic involvement or a clinical picture of multisystem involvement associated with histopathologic granulomatous inflammation. Early introduction of individualized immunosuppressive treatment may markedly improve the prognosis of the disease.
Bibliography
- Nadia Nathan , Pierre Marcelo , Véronique Houdouin , Ralph Epaud , Jacques de Blic , Dominique Valeyre , Anne Houzel et al- Lung sarcoidosis in children: update on disease expression and management, Thorax, 2015 Jun;70(6):537-42.doi: 10.1136/thoraxjnl-2015-206825.
- Abraham Gedalia , Tahir A Khan , Avinash K Shetty , Victoria R Dimitriades , Luis R Espinoza – Childhood sarcoidosis: Louisiana experience, Clin Rheumatol ,2016 Jul;35(7):1879-84. doi: 10.1007/s10067-015-2870-9
- Zletni MA, Abeed AM, Kawaja ES. Early-onset childhood sarcoidosis, manifesting as juvenile idiopathic arthritis: A case report. MOJ Orthop & Rheumatol. 2016;6:00234. doi: 10.15406/mojor.2016.06.00234.
- Majumder, Parthopratim Dutta; Biswas, Jyotirmay – Pediatric uveitis-An update, Oman Journal of Ophthalmology 6(3):p 140-150, Sep–Dec 2013. | DOI: 10.4103/0974-620X.122267
- Carine H Wouters, Anne Maes , Kevin P Foley ,John Bertin ,Carlos D Rose- Blau Syndrome, the prototypic auto-inflammatory granulomatous disease, Pediatric Rheumatology ,2014, 12,33
- Kousuke Fukuhara 1 , Aika Fukuhara, Jun Tsugawa, Shinji Oma, Yoshio Tsuboi– Radiculopathy in patients with Heerfordt’s syndrome: two case presentations and review of the literature, Brain Nerve , 2013 Aug;65(8):989-92.
- C Sileo , R Epaud, M Mahloul, N Beydon, D Elia, A Clement, H Ducou Le Pointe – Sarcoidosis in children: HRCT findings and correlation with pulmonary function tests, Pediatr Pulmonol, 2014 Dec;49(12):1223-33.doi: 10.1002/ppul.22956.
- Thomas El Jammal ,Yvan Jamilloux ,Mathieu Gerfaud-Valentin ,Dominique Valeyre ,Pascal Sève- Refractory Sarcoidosis: A Review, Therapeutics and Clinical Risk Management 2020:16 323–345
- Brian Chiu , Jackie Chan , Sumit Das , Zainab Alshamma ,Consolato Sergi-Pediatric Sarcoidosis: A Review with Emphasis on Early Onset and High-Risk Sarcoidosis and Diagnostic Challenges, Diagnostics 2019, 9, 160; doi:10.3390/diagnostics9040160
- Hasib Ahmadzai, Wei Sheng Joshua Loke, Shuying Huang, Cristan Herbert, Denis Wakefield,Paul S Thomas-Biomarkers in sarcoidosis: a review, Current Biomarker Findings 2014:4 93–106
- Simon Chauveau , Florence Jeny , Marie-Emeline Montagne , Rola Abou Taam 4
Véronique Houdouin , Ulrich Meinzer et al-Child–Adult Transition in Sarcoidosis: A Series of 52 Patients, J. Clin. Med. 2020, 9, 2097; doi:10.3390/jcm9072097