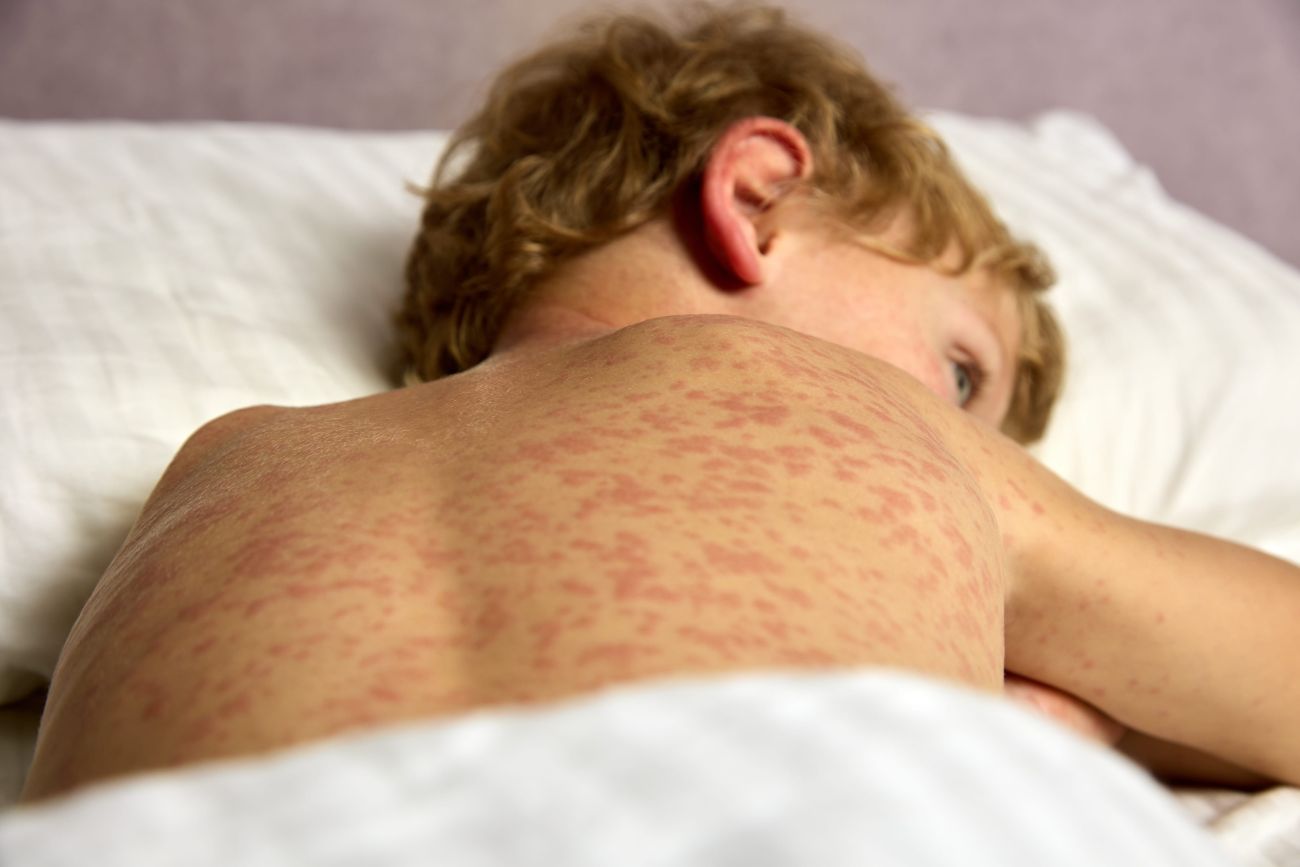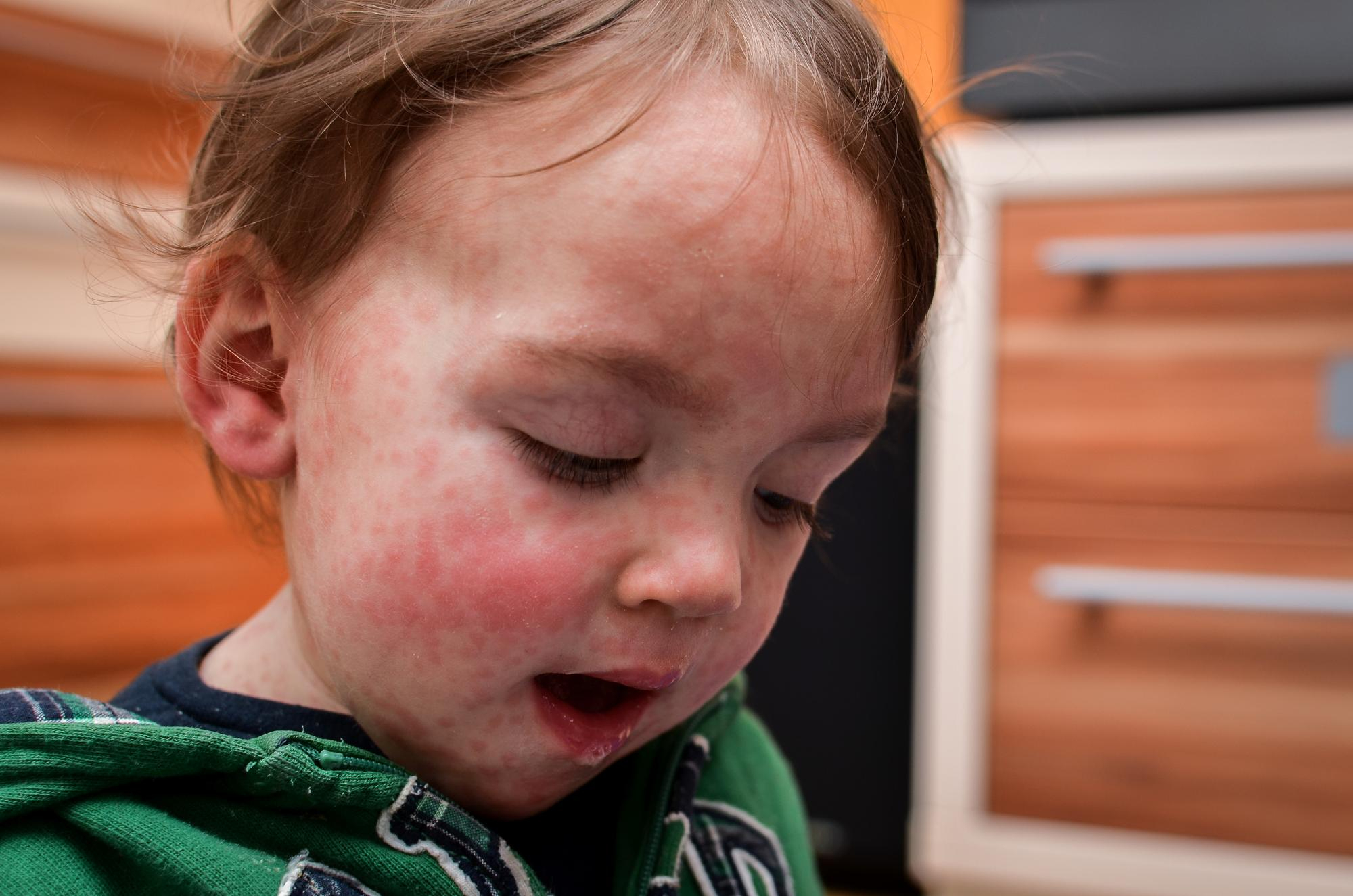
Kawasaki disease is a systemic vasculitis affecting medium-sized arteries, primarily in young children under five years of age. It is the leading cause of acquired heart disease in children in developed countries due to its association with coronary artery dilation and aneurysms. Recent advancements in knowledge have led to updates in the recommendations for the diagnosis and treatment of Kawasaki disease. This text highlights the most significant changes.
The most widely accepted diagnostic criteria for Kawasaki disease are those established by the American Heart Association (AHA). Additionally, the Japanese Ministry of Health Research Committee has its own set of criteria.
Until recently, the diagnostic criteria for the classic form of the disease required fever lasting at least five days, along with at least four of the following five symptoms:
– Bilateral conjunctival injection without discharge (Figure 1)
– Changes in the oral mucosa (such as chapped lips, a raspberry tongue, or a red pharynx) (Figure 2)
– Changes in the extremities (including redness of the palms and soles, edema of the hands and feet in the acute phase, and peeling of the skin around the nails after the acute phase)
– Polymorphic rash
– Cervical lymphadenopathy (with lymph nodes measuring at least 1.5 cm in diameter)
In the 2017 diagnostic guidelines for Kawasaki disease, the AHA acknowledged the ongoing debate regarding the minimum fever duration. It introduced a note stating that Kawasaki disease could be diagnosed in patients with fever lasting at least four days if they also exhibited at least four of the five cardinal symptoms. This was particularly emphasized in cases where there was evidence of redness or swelling of the palms or soles, or edema of the hands and feet.
In the 2024 revised diagnostic and treatment guidelines, fever lasting at least four days is now a mandatory criterion rather than an optional one (Table 1). However, an additional provision states that experienced clinicians may diagnose the disease earlier, after just three days of fever.
Table 1. Revised AHA diagnostic criteria for classic Kawasaki disease.
| Fever | Duration of 4 or more days with 4 out of 5 of the following: |
| 1. Conjunctivitis | Bilateral, bulbar conjunctival injection without exudate |
| 2. Lymphadenopathy | Cervical, >1.5 cm, usually unilateral |
| 3. Rash | Maculopapular, diffuse erythroderma or erythema multiforme |
| 4. Changes in the lips and oral mucosa | Red, chapped lips, strawberry tongue, or diffuse erythema of the pharynx |
| 5. Changes in the extremities | Erythema or edema of the palms and soles in the acute phase and periungual peeling of the skin in the subacute phase |
Unlike the criteria established by the AHA, the presence of fever for at least four days is not considered a mandatory criterion in the Japanese guidelines (Table 2). In other words, individuals who exhibit all five cardinal symptoms but are either afebrile or have a fever lasting fewer than four days (1, 2, or 3 days) can still be diagnosed with Kawasaki disease based on the Japanese criteria. Additionally, according to these guidelines, patients with four cardinal symptoms can be diagnosed with Kawasaki disease if echocardiography or coronary angiography reveals coronary aneurysm or dilation.
Table 2. Diagnostic criteria for Kawasaki disease as established by the Japanese Ministry of Health Research Committee.
| Main symptoms (at least 5 of the following) | |
| 1. Fever | Duration of 5 or more days |
| 2. Conjunctivitis | Bilateral |
| 3. Changes in the oral mucosa | Diffuse injection, strawberry tongue or chapped lips |
| 4. Rash | |
| 5. Changes in the extremities | Redness and swelling of the palms and soles; convalescent phase: peeling of the skin on the fingertips |
| 6. Acute nonsuppurative cervical lymphadenopathy | More than 1.5 cm in diameter |

Figure 1 Bulbar conjunctival injection without exudate sparing the limbus.

Figure 2 Red lips and strawberry tongue.
Other clinical symptoms that may be present but are not included in the main diagnostic criteria include arthritis, gastrointestinal involvement, irritability, lethargy, neurological manifestations, cough, and rhinorrhea. Kawasaki disease should also be considered in the differential diagnosis of infants and children with fever lasting more than seven days and unexplained aseptic meningitis.
In countries where the Bacillus Calmette-Guérin (BCG) vaccine is routinely administered, approximately 50% of Kawasaki disease patients may develop induration, erythema, or crusting around the BCG vaccination site (Figure 3). These findings can help support the diagnosis, particularly in children younger than three years of age. Induration around the BCG vaccination site is a distinctive clinical feature of Kawasaki disease, though it is not included in the five main criteria of either the AHA or Japanese guidelines. Even when patients exhibit four or fewer of the five major clinical criteria, physicians should be aware that a BCG vaccination site reaction may indicate Kawasaki disease.

Figure 3 Induration and erythema around the BCG vaccination site.
Between 10% and 15% of patients with echocardiographically confirmed Kawasaki disease do not meet the standard diagnostic criteria. This subset of patients is often classified as having “incomplete” or “atypical” Kawasaki disease, particularly in infants and children older than five years. The AHA has established guidelines to aid in diagnosing incomplete Kawasaki disease, and these criteria have remained unchanged to this day.
Patients with incomplete Kawasaki disease must meet all four of the following criteria:
- Fever lasting five days or more
- Presence of at least two major clinical symptoms of Kawasaki disease
- C-reactive protein (CRP) level greater than 30 mg/L or erythrocyte sedimentation rate (ESR) greater than 40 mm/h
- At least three additional laboratory criteria, which include:
- Anemia for age
- Platelet count ≥450 × 10⁹/L after day seven of fever
- Albumin level ≤30 g/L
- Elevated alanine transaminase
- Leukocyte count ≥15 × 10⁹/L
- Urine leukocytes ≥10 per high-power field
- A positive echocardiogram
Additionally, in patients with fever lasting five days or more and two or three major symptoms of Kawasaki disease, clinical and laboratory monitoring every other day is recommended if the fever persists. Moreover, if periungual scaling of the fingers and toes is observed, an additional cardiac ultrasound is advised.
Kawasaki shock syndrome and macrophage activation syndrome
In 2009, a distinct and severe form of Kawasaki disease known as Kawasaki shock syndrome (KSS) was identified. This under-recognized condition occurs in an estimated 5–7% of children with Kawasaki disease and is characterized by systolic hypotension or clinical signs of poor perfusion due to cardiogenic and/or distributive shock. It presents with reduced capillary refill, with or without left ventricular dysfunction, and/or relative volume overload requiring vasoactive drug support.
Kawasaki shock syndrome is more likely to develop in females, in patients with early-onset or incomplete Kawasaki disease, and in those with more severe laboratory abnormalities. These include thrombocytopenia, elevated C-reactive protein (CRP), hyponatremia, elevated transaminases, metabolic acidosis, signs of consumptive coagulopathy, and hypoalbuminemia. This patient group often exhibits resistance to intravenous immunoglobulin (IVIG) therapy, frequently requiring a second dose or additional treatments such as glucocorticoids, infliximab, or anakinra. They are also at a higher risk of coronary artery involvement, mitral regurgitation, and prolonged myocardial dysfunction. Early recognition of this complication and prompt initiation of appropriate treatment are crucial to ensuring a favorable outcome.
Macrophage activation syndrome (MAS) is another serious, potentially life-threatening complication of rheumatic diseases. It is characterized by excessive activation and proliferation of T lymphocytes and macrophages, leading to hemophagocytic activity and a hyperinflammatory state. This condition is associated with cytopenia, liver dysfunction, and a coagulopathy resembling disseminated intravascular coagulation. Extreme hyperferritinemia is a hallmark feature.
The incidence of MAS in Kawasaki disease is estimated to be 1.1–1.9%. Currently, there are no standardized criteria for diagnosing MAS in Kawasaki disease (Table 3). The most commonly used diagnostic criteria for MAS are those established by Ravelli et al. in patients with systemic juvenile idiopathic arthritis (sJIA-MAS). The HLH-2004 criteria are also occasionally used. While specific criteria for MAS in Kawasaki disease are lacking, some experts suggest that the 2016 sJIA-MAS criteria may be more applicable due to their higher sensitivity and specificity compared to the HLH-2004 criteria. Moreover, the parameters included in the 2016 sJIA-MAS criteria can be easily measured in blood tests at relatively low cost and are widely available, making them useful for MAS screening. Notably, splenomegaly is not included in the 2016 sJIA-MAS criteria because it is commonly observed in sJIA.
Table 3. The most frequently used criteria for the diagnosis of MAS
| 2016 sJIA-MAS | HLH-2004 (5/8 criteria) |
| A febrile patient with known or suspected sJIA is classified as MAS if the patient has:ferritin > 684 ng/mLand at least 2 of the 4 criteria listed: | Fever |
| Relative decrease in platelet count < 180 x 109/L | Splenomegaly |
| Elevated AST > 50 U/L | Cytopenia in >2 cell lines |
| Hypertriglyceridemia > 1.8 mmol/L | Triglycerides > 265 mg/dL or fibrinogen < 150 mg/dL |
| Hypofibrinogenemia < 3.6 g/L | Ferritin > 500 ng/ml |
| Soluble IL-2 receptor > 2400 U/mL | |
| Hemophagocytosis in the bone marrow, liver, or lymph nodes | |
| Decreased or absent NK cell activity |
Underdiagnosis of MAS in patients with Kawasaki disease remains a significant challenge. Distinguishing MAS from severe forms of Kawasaki disease, such as IVIG-resistant cases, can be difficult because both conditions share features such as persistent fever, elevated aminotransferase levels, thrombocytopenia, anemia, and hyperferritinemia. Some researchers suggest that resistant forms of Kawasaki disease may represent subclinical MAS. Notably, MAS is characterized by higher ferritin and triglyceride levels, more profound cytopenia, and hemorrhagic manifestations compared to refractory Kawasaki disease. This suggests that resistant forms of Kawasaki disease could represent an early stage of MAS.
Because MAS and refractory Kawasaki disease share a similar severe phenotype, and since multiple studies indicate that MAS should be considered in Kawasaki disease patients at high risk for IVIG resistance, routine laboratory screening for MAS is recommended in younger children, males, and those with elevated markers of inflammation.
New biomarkers that may aid in the diagnosis of Kawasaki disease
Several inflammatory markers, including erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), leukocyte count, and platelet count, can aid in the diagnosis of Kawasaki disease, either in combination with clinical symptoms or independently. However, these markers are generally nonspecific and may also be elevated in other conditions involving infection, inflammation, or autoimmunity.
Previous studies have shown that Kawasaki disease is associated with increased expression of various proinflammatory cytokines, including IL-6, IL-12, TNF-alpha, CXCL10, and interferon-gamma. However, routine measurement of these cytokines has not yet been implemented in clinical practice. B-type natriuretic peptide (BNP) and its inactive cleavage product, N-terminal prohormone brain natriuretic peptide (NT-proBNP), are being explored as potential biomarkers for Kawasaki disease. Several studies have demonstrated that NT-proBNP has high sensitivity and specificity in differentiating Kawasaki disease from other febrile illnesses. However, variations in NT-proBNP levels based on age make it difficult to establish a definitive cutoff value for distinguishing Kawasaki disease from other conditions.
MicroRNAs (miRNAs) are short, non-coding RNA molecules involved in gene expression regulation. Among them, miR-233 has been identified as one of the most highly expressed in the serum of Kawasaki disease patients. While identifying a single miRNA may be insufficient, analyzing the complete miRNA expression profile may improve the ability to differentiate Kawasaki disease from other febrile illnesses. In some countries, miRNA panels are already being used to aid in Kawasaki disease diagnosis.
Higher eosinophil expression, which may increase further after IVIG treatment, has been associated with a favorable response to IVIG therapy in Kawasaki disease. This suggests a potential protective role for eosinophils. Some studies have highlighted the importance of eosinophil levels in distinguishing Kawasaki disease from other febrile illnesses, indicating that eosinophils could be considered as an independent factor in future diagnostic criteria for Kawasaki disease.
Innovations in the Treatment of Kawasaki Disease
The treatment of Kawasaki disease follows recommendations from the American Heart Association (AHA) and the SHARE initiative (Single Hub and Access Point for Pediatric Rheumatology in Europe). The AHA guidelines were revised in 2024, while the SHARE recommendations were established in 2019.
Changes in Initial Treatment Recommendations
A key update in the 2024 AHA recommendations is the removal of routine initial treatment with IVIG and acetylsalicylic acid (ASA) for all patients. Instead, before initiating treatment, patients should be categorized into two groups based on their risk for coronary artery aneurysm development:
- Standard-risk patients
- High-risk patients
Patients in the high-risk category should receive more intensive initial therapy involving additional medications. High-risk patients include:
- Infants younger than six months
- Patients with a right coronary artery or left anterior descending coronary artery Z-score ≥ 2.5 on their initial echocardiogram
- Patients with a Son scoring system score of ≥ 3 (Table 4)
Table 4 Son scoring system for differentiating patients with Kawasaki disease who are at standard and high risk for developing coronary artery aneurysms
| Parameter | Number of points |
| Maximum Z-score on initial cardiac ultrasound ≥ 2 | 2 |
| Patient age at onset of fever <6 months | 1 |
| Asian origin | 1 |
| Initial CRP ≥ 13 mg/dL | 1 |
Interpretation: 0-1 (low risk), 2 (moderate risk), 3-5 (high risk).
For standard-risk patients, the AHA maintains its previous initial treatment recommendations, while the SHARE guidelines continue to recommend the same first-line treatment for all patients, without risk stratification.
The first-line treatment consists of:
- IVIG (2 g/kg, infused over 8–12 hours)
- Acetylsalicylic acid (ASA)
- The SHARE guidelines recommend 30–50 mg/kg/day in three or four divided doses.
- The AHA guidelines allow for a higher initial dose (80–100 mg/kg/day) until the patient becomes afebrile, after which the dose is reduced to 3–5 mg/kg/day.
For high-risk patients, a third medication is now recommended in addition to IVIG and ASA:
- Systemic glucocorticoids (prednisolone or methylprednisolone, 2 mg/kg)
- OR a TNF-alpha blocker (infliximab [single dose] or etanercept [three doses])
This addition represents the most significant innovation in Kawasaki disease treatment.
Management of IVIG-Resistant Kawasaki Disease
For patients who do not respond to the initial dose of IVIG (i.e., they remain febrile 36 hours after completing IVIG infusion), the 2024 AHA recommendations have changed treatment priorities.
Key updates:
- A second dose of IVIG is no longer preferred. Instead, alternative options should be considered, including:
- Systemic glucocorticoids
- TNF-alpha blockers (infliximab or etanercept)
- Interleukin-1 inhibitor (anakinra)
- Cyclosporine
- Cyclophosphamide (only for giant aneurysms)
- The SHARE recommendations allow for a second IVIG dose at the physician’s discretion but prioritize other immunosuppressive therapies if inflammation persists despite IVIG, glucocorticoids, and ASA.
Treatment of Kawasaki Shock Syndrome
The revised AHA guidelines also include recommendations for Kawasaki shock syndrome due to its high risk of IVIG resistance and coronary artery abnormalities.
- Patients with Kawasaki shock syndrome should receive intensified therapy from the start, including:
- IVIG
- ASA
- An additional drug (either a systemic glucocorticoid or a TNF-alpha blocker)
Management of Coronary Artery Aneurysms
The AHA provides specific recommendations for patients with coronary artery aneurysms:
- Medium-sized aneurysms (Z-score >5 but <10): Consider adding clopidogrel to low-dose ASA.
- Large aneurysms (Z-score ≥10 or internal diameter ≥8 mm):
- Introduce anticoagulation therapy in addition to ASA and clopidogrel.
- Newer direct oral anticoagulants (DOACs) may be a safer alternative to warfarin or low-molecular-weight heparin.
- All patients with aneurysms should receive additional anti-inflammatory therapy.
Management of MAS in Kawasaki Disease
The goal of MAS treatment is to rapidly suppress systemic inflammation and prevent organ damage. MAS is associated with thrombocytopenia, which may increase the risk of coronary artery aneurysms. However, there are no formal guidelines for MAS management in Kawasaki disease.
Current treatment strategies include:
- High-dose glucocorticoids (30 mg/kg/day for three days, then reduced to 2 mg/kg/day)
- If inadequate response to glucocorticoids:
- Parenteral cyclosporine A (reduces symptoms within 12–24 hours)
- IL-1 inhibitor (anakinra), which has been effective in treating MAS and systemic juvenile idiopathic arthritis (sJIA)
There is concern about overtreatment with potent immunosuppressive drugs, as MAS in Kawasaki disease often responds well to immunomodulatory therapy without requiring cytotoxic agents that can cause hepatic and renal complications. Reports suggest that IL-1 blockade with anakinra has been effective in treating refractory Kawasaki disease complicated by MAS, with many patients achieving positive outcomes using immunomodulators rather than intensive chemotherapy.
Summary of Key Updates:
- Risk stratification before treatment initiation (standard-risk vs. high-risk patients).
- A second dose of IVIG is no longer preferred for IVIG-resistant patients.
- For high-risk patients, initial therapy should include a third drug (glucocorticoid or TNF-alpha blocker).
- KSS should be treated with intensified therapy immediately.
- New recommendations for managing coronary artery aneurysms (clopidogrel, anticoagulation, DOACs).
- MAS treatment prioritizes high-dose glucocorticoids, cyclosporine, and IL-1 inhibitors while avoiding overtreatment.
References:
Ayusawa M, Sonobe T, Uemura S, Ogawa S, Nakamura Y, Kiyosawa N et al.; Kawasaki Disease Research Committee. Revision of diagnostic guidelines for Kawasaki disease (the 5th revised edition). Pediatr Int. 2005;47(2):232-4.
Chen Y, Ding YY, Ren Y, Cao L, Xu QQ, Sun L et al. Identification of differentially expressed microRNAs in acute Kawasaki disease. Mol Med Rep. 2018;17(1):932-938.
Corinaldesi E, Fabi M, Scalabrini I, Praticò ER, Andreozzi L, Torcetta F et al. Kawasaki Disease Complicated with Macrophage Activation Syndrome: The Importance of Prompt Diagnosis and Treatment–Three Case Reports. Rheumato. 2023;3(4):201-209.
de Graeff N, Groot N, Ozen S, Eleftheriou D, Avcin T, Bader-Meunier B et al. European consensus-based recommendations for the diagnosis and treatment of Kawasaki disease – the SHARE initiative. Rheumatology (Oxford). 2019;58:672-82.
Fukushige J, Takahashi N, Ueda Y, Ueda K. Incidence and clinical features of incomplete Kawasaki disease. Acta Paediatr. 1994;83(10):1057-60.
Gamez-Gonzalez LB, Moribe-Quintero I, Cisneros-Castolo M, Varela-Ortiz J, Muñoz-Ramírez M, Garrido-García M, Yamazaki-Nakashimada M. Kawasaki disease shock syndrome: Unique and severe subtype of Kawasaki disease. Pediatr Int. 2018;60(9):781-790.
Han SB, Lee SY, Jeong DC, Kang JH. Should 2016 Criteria for Macrophage Activation Syndrome be applied in children with Kawasaki disease, as well as with systemic-onset juvenile idiopathic arthritis? Ann Rheum Dis. 2016;75(7):e44.
Henderson LA, Cron RQ. Macrophage Activation Syndrome and Secondary Hemophagocytic Lymphohistiocytosis in Childhood Inflammatory Disorders: Diagnosis and Management. Paediatr Drugs. 2020;22(1):29-44.
Henter JI, Horne A, Aricó M, Egeler RM, Filipovich AH, Imashuku S et al. HLH-2004: Diagnostic and therapeutic guidelines for hemophagocytic lymphohistiocytosis. Pediatr Blood Cancer. 2007;48(2):124-31.
Jone PN, Tremoulet A, Choueiter N, Dominguez SR, Harahsheh AS, Mitani Y et al.; American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee of the Council on Lifelong Congenital Heart Disease and Heart Health in the Young; Council on Cardiovascular and Stroke Nursing; Council on Cardiovascular Radiology and Intervention; and Council on Clinical Cardiology. Update on Diagnosis and Management of Kawasaki Disease: A Scientific Statement From the American Heart Association. Circulation. 2024;150(23):e481-e500.
Kanegaye JT, Wilder MS, Molkara D, Frazer JR, Pancheri J, Tremoulet AH et al. Recognition of a Kawasaki disease shock syndrome. Pediatrics. 2009;123(5):e783-9.
Kuo HC. Diagnosis, Progress, and Treatment Update of Kawasaki Disease. Int J Mol Sci. 2023;24(18):13948.
Lin KH, Chang SS, Yu CW, Lin SC, Liu SC, Chao HY et al. Usefulness of natriuretic peptide for the diagnosis of Kawasaki disease: a systematic review and meta-analysis. BMJ Open. 2015;5(4):e006703.
Liu X, Zhou K, Hua Y, Wu M, Liu L, Shao S et al. Neurological involvement in Kawasaki disease: a retrospective study. Pediatr Rheumatol Online J. 2020;18(1):61.
Liu XP, Huang YS, Xia HB, Sun Y, Lang XL, Li QZ et al. A Nomogram Model Identifies Eosinophilic Frequencies to Powerfully Discriminate Kawasaki Disease From Febrile Infections. Front Pediatr. 2020;8:559389.
McCrindle BW, Rowley AH, Newburger JW, Burns JC, Bolger AF, Gewitz M et al. American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee of the Council on Cardiovascular Disease in the Young; Council on Cardiovascular and Stroke Nursing; Council on Cardiovascular Surgery and Anesthesia; and Council on Epidemiology and Prevention. Diagnosis, Treatment, and Long-Term Management of Kawasaki Disease: A Scientific Statement for Health Professionals From the American Heart Association. Circulation. 2017;135:e927-e999.
Muise A, Tallett SE, Silverman ED. Are children with Kawasaki disease and prolonged fever at risk for macrophage activation syndrome? Pediatrics. 2003;112(6 Pt 1):e495.
Ravelli A, Minoia F, Davì S, Horne A, Bovis F, Pistorio A et al.; Paediatric Rheumatology International Trials Organisation; Childhood Arthritis and Rheumatology Research Alliance; Pediatric Rheumatology Collaborative Study Group; Histiocyte Society. 2016 Classification Criteria for Macrophage Activation Syndrome Complicating Systemic Juvenile Idiopathic Arthritis: A European League Against Rheumatism/American College of Rheumatology/Paediatric Rheumatology International Trials Organisation Collaborative Initiative. Arthritis Rheumatol. 2016;68(3):566-76.
Schulert GS, Grom AA. Macrophage activation syndrome and cytokine-directed therapies. Best Pract Res Clin Rheumatol. 2014;28(2):277-92.
Son MBF, Gauvreau K, Tremoulet AH, Lo M, Baker AL, de Ferranti S et al. Risk Model Development and Validation for Prediction of Coronary Artery Aneurysms in Kawasaki Disease in a North American Population. J Am Heart Assoc. 2019;8(11):e011319.
Uehara R, Igarashi H, Yashiro M, Nakamura Y, Yanagawa H. Kawasaki disease patients with redness or crust formation at the Bacille Calmette-Guérin inoculation site. Pediatr Infect Dis J. 2010;29(5):430-3.
Wang Y, Wang W, Gong F, Fu S, Zhang Q, Hu J et al. Evaluation of intravenous immunoglobulin resistance and coronary artery lesions in relation to Th1/Th2 cytokine profiles in patients with Kawasaki disease. Arthritis Rheum. 2013;65(3):805-14.