Introduction
Thrombotic microangiopathy (TMA) and antiphospholipid antibody syndrome (APLA, also called antiphospholipid syndrome or APS) are distinct thrombotic disorders that can affect children with severe, life-threatening outcomes. TMA is marked by microangiopathic hemolytic anemia, thrombocytopenia, and organ ischemia caused by microthrombi in capillaries and arterioles. APLA is an autoimmune condition that increases the risk of blood clots, with thrombotic events (either arterial or venous) occurring in the presence of persistent antiphospholipid antibodies. Both conditions are rare in children but need quick recognition and treatment. This summary highlights pediatric-specific epidemiology, clinical signs, diagnostic methods, management strategies, and when to involve pediatric rheumatology and other specialists to guide frontline inpatient care providers.
Thrombotic Microangiopathy in Children
Epidemiology and Clinical Presentation
TMA in children is most commonly caused by hemolytic uremic syndrome (HUS). Typical HUS (Shiga toxin-producing E. coli-associated HUS, STEC-HUS) accounts for the vast majority of pediatric cases (approximately 90%). It primarily affects young children, with a peak incidence in those under 5 years old and a median age of around 3 years, often following a prodrome of bloody diarrhea. Streptococcus pneumoniae-associated hemolytic-uremic syndrome (Sp-HUS) is the leading cause of atypical, non-diarrheal hemolytic-uremic syndrome (HUS) in children, accounting for approximately 5% of cases, and typically occurs in conjunction with invasive pneumococcal infections (e.g., pneumonia or meningitis). Complement-mediated HUS (atypical HUS) and other rare genetic causes (e.g., cobalamin metabolism defects, DGKE mutations) account for the remaining approximately 5% of pediatric HUS. Thrombotic thrombocytopenic purpura (TTP), caused by ADAMTS13 deficiency, is exceedingly rare in childhood, with an annual incidence of about 0.2 per million. When TTP occurs in pediatrics, it typically presents during adolescence, with childhood-onset cases accounting for less than 10% of all TTP cases.
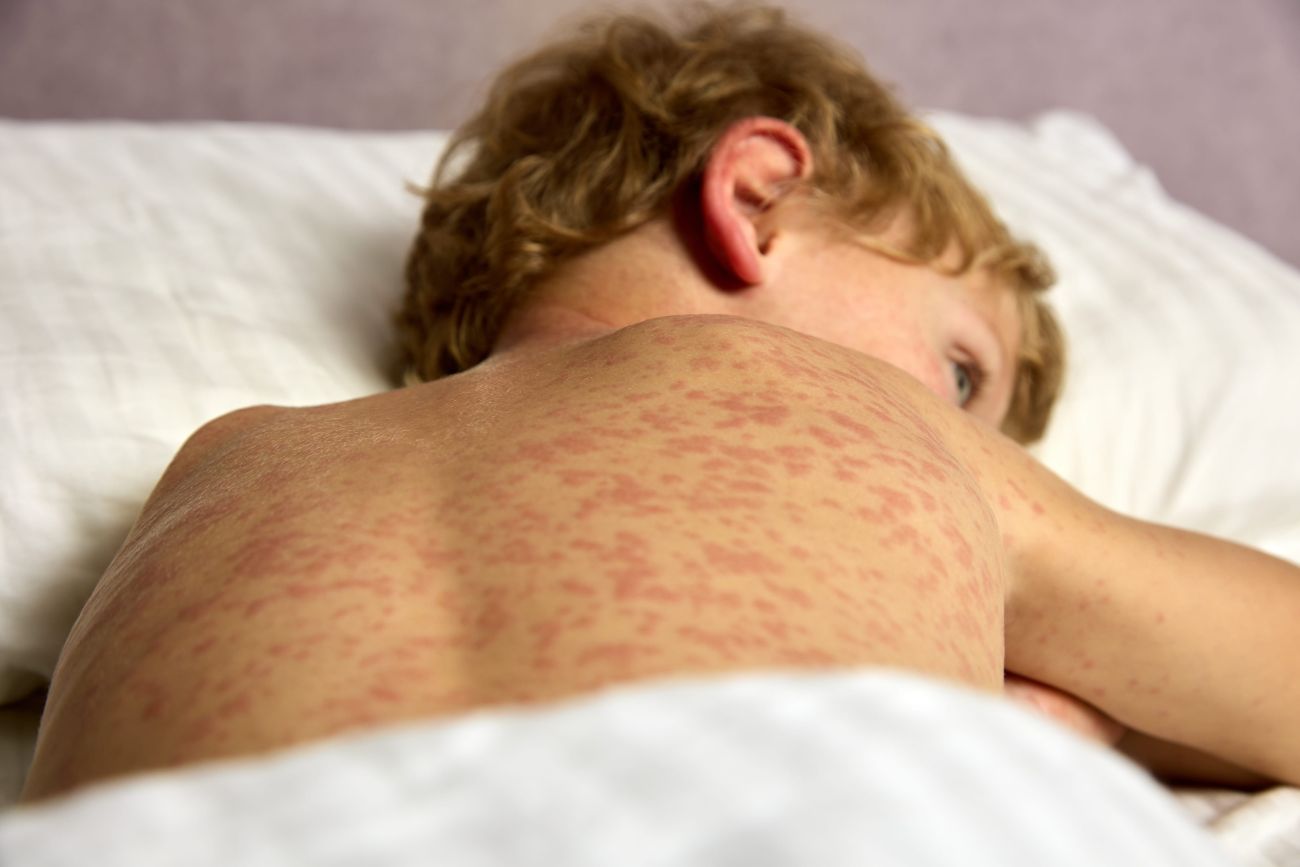
Clinically, pediatric TMA typically presents with the triad of microangiopathic hemolytic anemia (MAHA), thrombocytopenia, and organ injury (commonly acute kidney injury). Children with STEC-HUS often exhibit fatigue, pallor, and petechiae/bruising a few days after a diarrheal illness, progressing to oliguria or edema due to acute renal failure. Neurologic symptoms (seizures, altered mental status) are less common in HUS but can be a red flag for TTP. TTP classically features more pronounced neurologic involvement (headache, confusion, stroke, seizures) and less severe renal failure. Fever may occur in TTP, but the complete “pentad” (MAHA, thrombocytopenia, AKI, fever, neurologic signs) is often incomplete. Sp-HUS should be considered in a young child with pneumococcal sepsis or pneumonia who develops hemolysis and renal failure. Transplant-associated TMA is another context—children who have undergone hematopoietic stem cell or solid organ transplants can develop TMA as a complication, typically while inpatient.
Diagnostic Workup and Differential Diagnosis
The initial evaluation of a child with suspected thrombotic microangiopathy (TMA) should prioritize confirming the microangiopathic process and urgently distinguishing between the various etiologies of TMA. Key tests include a complete blood count (expecting anemia and thrombocytopenia), a peripheral blood smear (schistocytes and fragmenting RBCs confirm MAHA), hemolysis labs (elevated lactate dehydrogenase, low haptoglobin, elevated indirect bilirubin, and a negative direct Coombs test), and renal function tests (BUN and creatinine). Prompt evaluation for Shiga toxin-associated HUS is crucial—send stool culture or Shiga-toxin PCR if there is a history of diarrhea. If pneumonia or invasive pneumococcal infection is present, test for Streptococcus pneumoniae-related HUS (e.g., Coombs test often positive due to T-antigen exposure in Sp-HUS). ADAMTS13 activity should be sent urgently to evaluate for TTP; if the activity is very low (<10%) with an inhibitor, it supports the diagnosis of immune TTP. Complement studies (including C3 and C4 levels and screening for factor H autoantibodies) and genetic testing for mutations in complement regulatory genes may be indicated if atypical HUS is suspected. However, these results typically take longer to return. In critically ill cases, do not delay therapy while awaiting these confirmatory tests – instead, initiate presumptive treatment based on the most likely diagnosis (see Management).
Differential diagnosis of pediatric TMA includes other conditions that cause MAHA or thrombocytopenia:
- Disseminated intravascular coagulation (DIC) and thrombotic microangiopathy (TMA) can both lead to microangiopathic hemolysis and organ ischemia. DIC (often resulting from sepsis, malignancy, or trauma) is characterized by coagulopathy (prolonged PT/PTT, low fibrinogen, high D-dimer), which is typically not significant in primary TMA. In TMA, coagulation studies are generally normal or only mildly abnormal.
- Severe hypertension: Malignant hypertension or scleroderma renal crisis in children can produce microangiopathic changes and renal failure. Monitoring blood pressure is critical; hypertensive TMA improves with blood pressure control rather than plasma exchange or complement blockade.
- Autoimmune hemolysis with thrombocytopenia: Evans syndrome (autoimmune hemolytic anemia plus immune thrombocytopenia) may mimic thrombotic microangiopathy (TMA) with anemia and low platelet counts; however, a peripheral smear in Evans syndrome shows spherocytes without schistocytes, and the Coombs test is positive (unlike massive autoimmune hemolytic anemia, MAHA).
- Hemophagocytic lymphohistiocytosis (HLH) can cause cytopenias and organ failure, but is distinguished by extreme hyperferritinemia, fevers, and findings of hemophagocytosis – and typically does not cause macroangiopathic hemolytic anemia (MAHA) with schistocytes.
- Acute glomerulonephritis/vasculitis: Diseases such as acute glomerulonephritis, acute post-infectious glomerulonephritis, ANCA-associated vasculitis, or lupus nephritis can lead to renal failure, characterized by hematuria, proteinuria, and some hemolysis. They may even trigger secondary TMA, but primary features (e.g., immunologic markers, renal biopsy findings) help identify them.
- Others: Transfusion reactions or clotting disorders (like heparin-induced thrombocytopenia) could result in thrombocytopenia and hemolysis (HIT can cause thromboses with low platelets, but usually not MAHA). Metabolic disorders (e.g., inherited cobalamin C defect) can lead to recurrent TMA in infants – a history of failure to thrive or neurological issues may necessitate a metabolic workup.
In practice, distinguishing TTP from typical HUS is a critical early decision: TTP can be rapidly fatal without plasma exchange, whereas typical STEC-HUS is managed supportively. Clues favoring TTP include prominent neurologic symptoms, minimal diarrheal prodrome, and an older child or teen; clues for HUS include a recent diarrheal illness or known pneumococcal infection and predominant renal injury. If in doubt, empirically treat for TTP (with plasma exchange) while continuing the workup, since missing TTP can be fatal.
Laboratory Findings and Imaging Studies
Hematologic labs in TMA classically show evidence of microangiopathic hemolytic anemia: schistocytes on smear, elevated LDH, low haptoglobin, and a negative direct antiglobulin test (Coombs). Thrombocytopenia is typically significant (with platelets often <50–100×10^9/L). Coagulation tests are usually standard in primary TMA, which helps differentiate it from DIC. In pneumococcal HUS, the direct Coombs test may be positive due to polyagglutination (activation of the Thomsen-Friedenreich antigen on RBCs). Renal function tests typically reveal acute kidney injury in most HUS cases (elevated BUN/Cr, metabolic acidosis, and hyperkalemia in severe cases). Hematuria and proteinuria are generally present on urinalysis in HUS.
Specialized assays guide etiology: ADAMTS13 activity <10% confirms TTP (with an inhibitor test indicating acquired TTP). Complement levels (low C3, low CH50) can suggest complement-mediated aHUS, although normal levels do not exclude it; genetic panels for complement regulatory genes (CFH, CFI, MCP, etc.) or tests for anti-Factor H antibody can help identify the cause of aHUS, but these often return after acute management. Vitamin B12 levels or methylmalonic acid may be checked in infants to exclude metabolic causes of TMA (e.g., cobalamin C defect). If a systemic disease is suspected, relevant laboratory tests, such as ANA, anti-dsDNA (for SLE), or ANCA (for vasculitis), should be sent in parallel.
Imaging is guided by organ involvement. A renal ultrasound may demonstrate acute renal injury, but it is typically nonspecific, often showing enlarged and echogenic kidneys in acute kidney injury (AKI). If neurologic signs are present, neuroimaging (CT/MRI brain) is warranted to evaluate for stroke or hemorrhage. In TTP or HUS with severe hypertension, brain imaging can also assess for posterior reversible encephalopathy syndrome (PRES). In transplant-associated thrombotic microangiopathy (TMA), a biopsy of the transplanted organ (e.g., kidney) may reveal thrombi in the microvasculature. While not diagnostic, imaging findings can assist in assessing complications (e.g., cardiac echo for heart failure in severe hypertension or to detect cardiac thrombi in TTP). Ultimately, the diagnosis of TMA is laboratory-based; imaging serves as an adjunct to evaluate complications.
Acute Management and Treatment Strategies
Management of pediatric TMA must begin before confirming the cause, based on the primary clinical suspicion, as early intervention is critical. Key acute treatments include:
- Supportive care for all TMA: This includes aggressive blood pressure control, correction of fluid and electrolyte imbalances, and transfusion support. RBC transfusions are provided for severe anemia. Platelet transfusions are avoided in TTP unless a life-threatening hemorrhage occurs, as they can fuel thrombosis. In HUS, careful fluid management is crucial; many children require dialysis (peritoneal or hemodialysis) for AKI. Avoid nephrotoxic drugs and consider renal replacement early if needed.
- Plasma exchange (PLEX): If TTP is suspected (or if atypical HUS vs TTP cannot be immediately distinguished in a critically ill child), start daily plasma exchange promptly. PLEX removes autoantibodies against ADAMTS13 and replenishes the ADAMTS13 enzyme. High-dose corticosteroids (e.g., IV methylprednisolone) should be added concurrently for acquired TTP. Rituximab is often used in cytopenic purpura (of) and thrombotic thrombocytopenic purpura (en) to suppress antibodies in adolescents. Data on adolescents ‘ adolescence in pediatrics reflects adult practice. For hereditary TTP (congenital ADAMTS13 deficiency, also known as Upshaw-Schulman syndrome), plasma infusions (FFP) can be utilized as an alternative to plasma exchange, as there are no autoantibodies present; plasma provides the missing ADAMTS13.
- Eculizumab: For complement-mediated atypical HUS, eculizumab (a monoclonal antibody against C5) is the first-line therapy. If the clinical picture suggests aHUS (absence of diarrhea, negative Shiga testing, normal ADAMTS13 levels, evidence of complement dysregulation, or a family history), eculizumab should be initiated as soon as possible. Early eculizumab has dramatically improved outcomes in aHUS, halting thrombotic microangiopathy (TMA) and enhancing renal recovery. Eculizumab is typically continued every 2–3 weeks for at least several months; some patients may require long-term maintenance to prevent relapse. (Note: Ensure meningococcal vaccination before or concurrently with eculizumab due to infection risk.)
- Supportive care in STEC-HUS: There is no specific antidote for typical HUS; management is supportive. Antibiotics are generally avoided in acute diarrheal hemolytic-uremic syndrome (HUS) due to concerns about increasing toxin release, except in specific scenarios such as sepsis. Early dialysis for uremia or fluid overload, alongside transfusions as needed, are mainstays of treatment. Most children with STEC-HUS will recover renal function with supportive care, although a minority may experience residual kidney damage. In Sp-HUS, management is also broadly supportive (appropriate antibiotics for the pneumococcal infection plus intensive care support); some centers have successfully used eculizumab in severe Sp-HUS cases to control complement activation, but evidence is still evolving.
- Adjunct therapies: In adult TTP, caplacizumab (a vWF-cleaving protease modulator) is employed to mitigate microthrombi formation rapidly. Pediatric experience is limited but growing; case series indicate that caplacizumab is safe and may accelerate platelet count recovery in children with immune TTP. Its use in pediatric TTP can be considered in refractory or severe cases in consultation with hematology. For refractory complement TMA that does not respond to eculizumab, some experimental complement inhibitors or immunosuppressive strategies (e.g., plasma exchange to remove an autoantibody against complement factor H) may be considered.
Monitoring: Children with TMA should initially be in an intensive care unit (ICU) or a closely monitored setting. Follow daily labs (CBC, LDH, chemistries) to gauge hemolysis and organ function. Therapeutic plasma exchange is continued in TTP until platelet count and LDH normalize for at least 2 days and ADAMTS13 improves. For HUS, monitor fluid status and renal function; dialysis is weaned as kidney function returns.
Indications for Rheumatology Consultation and Multidisciplinary Care
Pediatric TMA often requires a team approach. Early involvement of nephrology is essential, particularly in HUS, for management of dialysis and long-term renal follow-up. Hematology is typically consulted for TTP or unexplained hemolytic anemia; they coordinate plasma exchange and specialized lab testing. Pediatric rheumatology should be engaged if an underlying autoimmune process is suspected or confirmed. For instance, TMA can occur in systemic lupus erythematosus (SLE) or antiphospholipid syndrome, and rheumatology will assist with managing immunosuppression if TMA is part of an autoimmune disease. In catastrophic APS, a rare and fulminant form of APS that causes multiorgan microthrombi, thrombotic microangiopathy (TMA) may be a feature; rheumatology’s input is crucial for immunomodulatory therapy in this context.
Additionally, rheumatology can help unravel complex cases, such as TMA with positive ANA or other autoimmune markers. Different specialists are brought in as needed: critical care for ICU support, neurology for CNS involvement (like stroke or seizures), and the transplant team for transplant-associated TMA. Multidisciplinary rounds are beneficial given the overlapping domains; for example, a child with atypical HUS may require the services of nephrologists for plasma exchange and dialysis, hematologists for authorization of eculizumab, and rheumatologists if the workup reveals an autoimmune etiology, such as complement dysregulation, that necessitates genetic counseling.
Prognosis and Follow-Up
- Outcomes in pediatric TMA have significantly improved with modern therapies; however, prognostic considerations vary depending on the etiology.
- STEC-HUS: Most children survive the acute episode, with mortality rates being low in experienced centers. Renal outcomes are generally good, though up to approximately 30% may have long-term kidney sequelae (hypertension, reduced GFR). End-stage renal disease is relatively uncommon (1–3% in modern series). All STEC-HUS patients need follow-up with nephrology to monitor blood pressure, kidney function, and urine protein, as chronic kidney disease can manifest later.
- Strep pneumo-HUS: Historically, it has had worse outcomes than typical HUS. Older reports noted acute mortality around 12% and long-term renal impairment in roughly 30% of survivors. However, recent data suggest that if meningitis is absent, mortality can be as low as a few percent. Nonetheless, Sp-HUS is still considered a severe disease. These patients require close follow-up for renal recovery; some may need prolonged dialysis or transplant evaluation if kidney failure persists. Recurrence of Sp-HUS is rare (related to new invasive pneumococcal infection), and appropriate pneumococcal vaccination is an essential preventive measure.
- Complement-mediated aHUS: Before complement inhibitors, aHUS carried a poor prognosis – the majority progressed to end-stage renal disease or died within a year of diagnosis. With eculizumab, outcomes have dramatically improved: TMA remission is achieved in most cases, and many children experience significant recovery of kidney function. The relapse rate is low while on therapy. However, discontinuing eculizumab can risk relapse, especially in those with specific genetic mutations. Long-term prognosis depends on kidney recovery; some children still progress to chronic kidney disease. Lifelong follow-up with nephrology is needed, and many remain on eculizumab (or the longer-acting agent ravulizumab) indefinitely to prevent relapses. Families benefit from genetic counseling, as aHUS mutations can be inherited.
- TTP: Untreated TTP is often fatal (>90% mortality), but with prompt plasma exchange therapy, survival now exceeds 80–90%. Pediatric outcomes are similar to those of adults when treated aggressively. Acquired immune TTP can relapse (especially in the first year or two); some centers preemptively use rituximab or maintenance immunosuppression to prevent relapses. Children with hereditary TTP require lifelong plasma infusions prophylactically (or newer therapies like recombinant ADAMTS13 when available) to prevent episodes. Neurological deficits from TTP (stroke, etc.) may cause lasting morbidity, so neurodevelopmental follow-up and rehabilitation services are essential for those with CNS involvement. All TTP survivors should be monitored for relapse signs and have a plan to check ADAMTS13 activity periodically.
- Other TMAs: For malignancy- or transplant-associated TMA, prognosis hinges on treating the underlying trigger (e.g., stopping a causative drug, treating rejection). These cases can have high morbidity due to the overall critical illness. Prognosis is guarded, and long-term follow-up is typically with the respective specialty (oncology or transplant team).
Across all pediatric TMAs, transitioning care as patients age is crucial. Pediatric patients with chronic TMA conditions (e.g., aHUS, hereditary TTP) will need to move to adult providers knowledgeable about these rare disorders. Early involvement of care coordination, patient education, and support groups (where available) can enhance long-term outcomes.
Antiphospholipid Antibody Syndrome in Children
Epidemiology and Clinical Presentation
Antiphospholipid antibody syndrome (APLA/APS) in children is a rare condition. While the incidence of APS in adults is approximately 2 per 100,000 per year, the number of pediatric APS cases is much smaller (exact incidence unknown). One large children’s hospital identified only 21 pediatric APS cases over nearly 20 years, highlighting its rarity. Pediatric APS can be primary (occurring on its own) or secondary to another disease. Secondary APS is more common in children, usually occurring in the context of childhood-onset systemic lupus erythematosus (SLE). In one case series, almost half of pediatric APS patients had a preceding lupus diagnosis. The median age of diagnosis is in adolescence (mid-to-late teens) – APS is exceedingly uncommon before puberty. However, antiphospholipid antibodies may be transiently detected in younger children (often after infections) without causing thrombosis.
Children with APS present clinically with thromboembolic events. Venous thromboembolism (VTE) is a common condition; deep vein thrombosis (DVT) of the extremities and pulmonary embolism can occur, sometimes triggered by factors such as the use of central venous lines or immobility. Arterial thromboses also occur in pediatric APS, particularly strokes. Ischemic stroke is a common presentation of APS in adolescents and even school-age children, often raising suspicion for an underlying aPL syndrome, especially if no conventional stroke risk factors are present. Other arterial events, such as myocardial infarction, are rare in children (except in complex cases or those with longstanding disease). Young children (<10 years) with APS are uncommon, but reported instances include renal vein thromboses or cerebral sinovenous thromboses, often associated with catastrophic APS or concomitant infections.
Beyond overt clots, children with APS frequently exhibit non-criteria manifestations that may alert providers. Up to two-thirds display features such as chronic thrombocytopenia, Coombs-positive hemolytic anemia, or livedo reticularis of the skin. Livedo reticularis (a mottled purplish vascular pattern on the skin) is a classic but non-specific finding that, in the presence of thrombosis, should raise concerns for APS. Other potential features can include migraines, seizures, or chorea (attributed to aPL-related neurovascular injury), as well as, in some cases, cardiac valve thickening or regurgitation (Libman-Sacks endocarditis), as observed in echocardiograms of lupus patients with aPL. Importantly, since pregnancy morbidity is one of the criteria for APS in adults, it’s important to note that adolescent girls with APS may have a history of unexplained fetal loss or pregnancy complications. Though this is rarely applicable in pediatrics, it may occur in older teens.
Catastrophic Antiphospholipid Syndrome (CAPS) is an acute, life-threatening variant in which multiple organ systems develop small-vessel thromboses within a short timeframe. It is scarce in children but has been reported. Children with CAPS present with multi-organ failure – for instance, simultaneous respiratory distress (pulmonary emboli), renal failure, liver impairment, CNS infarcts, and skin necrosis – often triggered by an underlying condition such as infection. CAPS has a high mortality rate and necessitates urgent therapy (discussed below).
Diagnostic Evaluation and Differential Diagnosis
Diagnosing APS in a child requires a high level of suspicion. Any child or adolescent with an unprovoked thrombosis or stroke, especially if autoimmune features such as rash or arthritis are present, should be evaluated for antiphospholipid syndrome. The revised Sapporo/Sydney classification criteria (used in adults) are also applied to children, requiring at least one clinical event (vascular thrombosis or pregnancy morbidity) plus persistent laboratory evidence of antiphospholipid antibodies on two separate occasions at least 12 weeks apart. Pregnancy-related criteria rarely apply to pediatric patients, so the primary focus is on thrombosis. Antiphospholipid antibody testing includes the lupus anticoagulant (LA) assay, anticardiolipin antibody (aCL, IgG and IgM), and anti-β2-glycoprotein I (anti-β2GPI, IgG and IgM). At least one of these should be positive at moderate to high titer or show strong positivity for diagnosis; low titers or transient positives are not sufficient. In children, these antibodies may temporarily rise after infections, especially viral illnesses, and then disappear. Therefore, a repeat positive test 12 weeks later is essential to confirm “true” APS rather than transient aPL positivity.
The workup for a hospitalized child with suspected APS centers on confirming the thrombosis and assessing for secondary causes. Key steps include:
- Imaging to confirm thrombosis: Utilize appropriate studies (e.g., Doppler ultrasound for lower extremity deep vein thrombosis, CT angiography for pulmonary embolism, MRI/MRA for cerebral thrombosis or stroke). In stroke cases, also assess intracardiac sources (such as an echocardiogram for cardiac thrombi or valve lesions) as part of the workup, as APS can lead to valvular disease or intracardiac thrombus.
- Antiphospholipid antibody panel: Send LA, aCL, and anti-β2GPI tests. Note that an acute-phase reaction or ongoing anticoagulation can affect some tests (e.g., lupus anticoagulant testing can be confounded by heparin or elevated C-reactive protein). If an APS diagnosis is strongly suspected but initial tests are negative, they should be repeated off anticoagulation and when the child is stable. Also, if initial testing is positive, remember to repeat it after 12 weeks to meet the criteria. However, do not delay treatment while awaiting repeat testing – a presumptive APS diagnosis can be made in the interim if clinical suspicion is high.
- Evaluation for underlying disease: Since many pediatric APS cases are related to lupus, test for ANA, dsDNA, and other lupus markers (such as complement levels). Similarly, inquire about and examine any other autoimmune symptoms (e.g., arthritis, rash, oral ulcers) that might indicate SLE. If lupus is confirmed, APS is considered secondary, and management must also address systemic lupus erythematosus (SLE).
- Thrombophilia workup: Consider screening for other prothrombotic conditions that might coexist or mimic APS, especially in cases of idiopathic clots. This may include assessing protein C/S and antithrombin levels, testing for Factor V Leiden or prothrombin gene mutations, and evaluating for hyperhomocysteinemia, among others. In practice, a comprehensive thrombophilia panel may not be performed acutely in all cases; however, in instances of unprovoked thrombosis, it is reasonable to check for common inherited factors. When dealing with adolescents, inquire about oral contraceptive use, as estrogen-containing birth control is a risk factor for clots and is contraindicated in APS.
- Differential diagnoses: When evaluating a child with thrombosis and positive aPL, consider:
- Provoked thrombosis: Many pediatric clots result from central venous catheters, trauma, surgery, or prolonged immobility. While the presence of such triggers does not rule out APS, idiopathic clots are more indicative. If a known strong provoking factor is present (e.g., an indwelling catheter), caution is warranted in labeling it as APS unless antibodies remain positive later.
- Inherited thrombophilias, such as Factor V Leiden or prothrombin gene mutations, can lead to thrombosis in teenagers; however, these do not typically cause antibody positivity. They may coexist, however.
- Heparin-induced thrombocytopenia (HIT): In hospitalized adolescents receiving heparin, HIT can lead to thromboses accompanied by thrombocytopenia; HIT antibodies should be tested if the timeline and exposure align. HIT would not account for positive aCL or β2GPI, though.
- Thrombotic microangiopathies: TMA (as discussed above) can result in multi-organ thromboses and may even produce a positive lupus anticoagulant (prolonged PTT) in some situations. If microangiopathic hemolysis is present, consider TTP/HUS versus catastrophic antiphospholipid syndrome (APS) as a differential diagnosis. Distinguishing CAPS from TTP can be challenging – CAPS often has a known background of APS or SLE and generally requires both anticoagulation and immunosuppression, whereas TTP focuses on plasma exchange.
- Other vasculopathies: Children with vasculitis (such as Kawasaki disease or polyarteritis nodosa) can develop thromboses or aneurysms, but their lab profile differs. COVID-19-related multisystem inflammatory syndrome (MIS-C) has been linked to thromboses and transient aPL positivity in some cases, but the complete APS picture (persistent antibodies, chronic risk) is typically absent.
- Provoked thrombosis: Many pediatric clots result from central venous catheters, trauma, surgery, or prolonged immobility. While the presence of such triggers does not rule out APS, idiopathic clots are more indicative. If a known strong provoking factor is present (e.g., an indwelling catheter), caution is warranted in labeling it as APS unless antibodies remain positive later.
In summary, diagnosing pediatric APS requires demonstrating the presence of antiphospholipid antibodies persistently and linking them to clinical thrombotic events. Due to its rarity, this diagnosis is often one of exclusion, made after ruling out other hypercoagulable causes and typically in consultation with a pediatric rheumatologist or hematologist who is familiar with antiphospholipid syndrome (APS).
Laboratory Studies and Imaging
Antiphospholipid antibody testing: The laboratory hallmark of antiphospholipid antibody syndrome (APLAS) is the presence of one or more antiphospholipid antibodies on at least two occasions. Tests include:
Lupus anticoagulant (LA): This is a functional coagulation assay (typically a dilute Russell viper venom test or PTT-based assay with phospholipid dependence). Paradoxically, LA prolongs phospholipid-dependent clotting times in vitro but causes thrombosis in vivo. In children with APS, LA is frequently positive; however, false positives can occur (e.g., in acute infections).
Anticardiolipin (aCL) antibodies: Measured by ELISA (IgG and IgM isotypes). Medium to high titers (>40 GPL or MPL, or above the 99th percentile) are considered positive according to the criteria. Low titers may be transient or not clinically significant.
Anti-β2 glycoprotein I antibodies are also measured by ELISA (IgG and IgM). These often correlate with aCL positivity, especially in autoimmune APS. High titers (again >99th percentile) are significant.
It’s important to note that many “positive aPL” results in children do not translate to accurate APS. Transient aCL or LA can occur following viral infections or even in up to 10% of healthy children transiently. Therefore, persistent positivity and appropriate clinical context are key. In practice, if a hospitalized child tests positive for an aPL after a thrombotic event, it is recommended to repeat the test 12 weeks later (as an outpatient) to confirm persistence while managing the thrombosis in the interim.
General laboratory workup: In pediatric APS, other laboratory tests reflect the consequences of thromboses or the underlying disease. For example, a child with APS secondary to lupus may exhibit anemia of chronic disease or hemolytic anemia, leukopenia, or thrombocytopenia (often mild). Thrombocytopenia is common in pediatric APS (though usually moderate, with platelet counts of 100-150×10^9/L). Markers of inflammation (ESR, CRP) might be elevated if an underlying inflammatory disease is active or if the thrombosis caused tissue injury. If there is concern for CAPS, DIC labs (fibrinogen, D-dimer, INR) can be very high because CAPS may resemble DIC.
Imaging for thromboses is critical for diagnosis and monitoring. Depending on the site, this may include: Doppler ultrasonography for deep vein thrombosis (DVT) or renal vein thrombosis in the extremities, echocardiography for intracardiac thrombi or valvular lesions, CT or MR angiography for the pulmonary or cerebral vessels, and MRI for brain ischemia. In children with strokes, brain MRI often shows ischemic infarcts in arterial territories; if related to APS, there may be evidence of prior silent infarcts or changes in white matter as well. Some children with recurrent APS may undergo surveillance imaging (e.g., periodic echocardiograms to monitor valvular disease progression or screening ultrasounds if at high risk for certain clots). However, this is assessed on a case-by-case basis.
Other studies: In secondary APS, tests about the primary disease (e.g., lupus serologies) will be positive and guide management. Antinuclear antibody (ANA) is positive in the majority of lupus-associated antiphospholipid syndrome (APS) patients. Additionally, the Direct Coombs test might be positive in those with autoimmune hemolysis (one of the non-criteria APS manifestations). Biopsies are rarely needed for diagnosis; however, in catastrophic APS, a biopsy of affected tissue (such as skin or kidney) can reveal small vessel thrombosis, which can support the diagnosis in ambiguous cases.
Acute Management and Treatment
Management of pediatric APS is primarily based on adult APS guidelines, with adaptations tailored to the pediatric context. Key principles include anticoagulation for thrombosis, avoidance of triggers that can lead to recurrence, and addressing any underlying autoimmune diseases.
Acute thrombosis treatment: In a child with APS and an acute thrombotic event, immediate anticoagulation is essential. Typically, unfractionated heparin or subcutaneous low-molecular-weight heparin (LMWH, e.g., enoxaparin) is initiated for the treatment of venous or arterial thrombosis. Heparin offers the advantage of quick reversibility and ease of monitoring, which is beneficial when there is a high risk of bleeding (e.g., large stroke with hemorrhagic transformation risk). LMWH is often preferred in children for its practicality and reliable dosing. Thrombolysis (tPA) is reserved for massive, life-threatening thromboses, such as massive pulmonary embolism (PE) with hemodynamic collapse or an acute ischemic stroke in a critical vessel, within the very early window. However, anticoagulation is generally sufficient for most APS-related clots.
After initial heparin treatment, the standard long-term therapy involves transitioning to a
vitamin K antagonist (warfarin) to maintain an INR of 2.0–3.0. Warfarin effectively prevents the recurrence of both venous and arterial events in APS and remains the standard of care for adults with APS. In pediatric patients, warfarin management poses challenges due to variable diets and the difficulty of frequent blood draws; however, it is feasible for older children and teens. In younger children or when warfarin is impractical, continuing LMWH injections or even aspirin (in some low-risk situations) have been utilized as alternatives. However, these may be less effective for high-risk APS. Ongoing research is examining direct oral anticoagulants (DOACs) in APS; current adult guidance advises against DOACs (like rivaroxaban) in high-risk APS (especially “triple-positive” patients) due to inferior outcomes. In children, the use of DOACs is not well studied in APS and is typically avoided in known APS. However, it may be considered on a case-by-case basis for single venous events with low-risk antibody profiles when warfarin is not feasible.
Secondary APS management: If a child has APS secondary to SLE or another autoimmune disease, managing that disease is vital. For lupus, this entails immunosuppression to control disease activity (e.g., corticosteroids, hydroxychloroquine, immunosuppressants). Hydroxychloroquine (HCQ) warrants special mention – in adults with APS (especially those with lupus), HCQ has been shown to provide protective anticoagulant and endothelial benefits, thereby reducing the risk of thrombosis. Pediatric rheumatologists similarly prescribe HCQ for children with lupus or aPL positivity, as it is low-risk and likely beneficial. Every child with SLE and antiphospholipid antibodies should be on HCQ unless contraindicated.
Catastrophic APS (CAPS) treatment: CAPS requires an aggressive combined approach often termed the “triple therapy”: 1) Anticoagulation (usually continuous IV heparin infusion); 2) High-dose corticosteroids (to dampen the cytokine storm and autoimmune response, e.g., methylprednisolone 1g daily IV pulses); and 3) Plasma exchange and/or IV immunoglobulin (IVIG) to remove or neutralize antiphospholipid antibodies. Additionally, cyclophosphamide is added if CAPS occurs in a lupus patient or if there is an underlying trigger, such as a flare. Newer adjuncts in refractory CAPS include rituximab (to deplete B-cells producing antibodies) and eculizumab (to halt complement-mediated microthrombosis seen in CAPS). In pediatric CAPS, case reports have shown some success with these approaches, but mortality remains high. Management should occur in an ICU with multidisciplinary input (rheumatology, hematology, critical care). Other adjunct measures: Low-dose aspirin is sometimes used alongside anticoagulation, particularly in arterial events or if there are recurrent miscarriages (in older teens). In children with only laboratory aPL positivity but no thrombosis (“aPL carriers”), prophylactic aspirin might be considered if they have other risk factors; however, generally, we do not anticoagulate until a thrombotic event has occurred. If a child with aPL is undergoing high-risk situations (surgery, prolonged immobilization), short-term prophylactic heparin or aspirin can be utilized to prevent provoked clots. Lifestyle modifications and preventive measures are essential for adolescents with APS. They should avoid smoking and estrogen-containing contraceptives (due to increased thrombotic risk). If contraception is needed, progestin-only methods or IUDs are recommended. Teens should also be counseled on maintaining a healthy weight and staying active (while avoiding dehydration on long flights, etc.), as these general measures help reduce the risk of thrombosis.
When to Consult Rheumatology and Multidisciplinary Care
Pediatric rheumatology should be involved early in cases of confirmed or suspected APS. Due to the overlap with lupus and other rheumatic diseases, rheumatologists can assist in interpreting antibody tests and guiding immunomodulatory treatment when necessary. They will also play a crucial role in the long-term care and monitoring of APS, particularly if immunosuppressive therapy or criteria for discontinuing anticoagulation are being considered. In an inpatient setting, consult rheumatology for any child with documented antiphospholipid antibodies and thrombosis, or for any child with thrombosis and features of autoimmune disease (even if APS is not yet confirmed). Rheumatology can help determine if the criteria are met and coordinate the necessary follow-up testing for aPL persistence.
Hematology is another essential consultant. Pediatric hematologists are experts in managing anticoagulation in children and often co-manage APS patients, as some centers view APS primarily as a hematologic disorder. Hematology consultation is essential for guidance on the use of warfarin versus low-molecular-weight heparin (LMWH), monitoring anticoagulation, and addressing specialized scenarios, such as concurrent thrombocytopenia or clotting disorders. In cases of catastrophic APS, hematology will be involved in managing plasma exchange and providing blood product support.
A multidisciplinary approach is critical for APS, which can affect virtually any organ. Depending on the manifestation, involve neurology for stroke management and neurorehabilitation, nephrology for APS nephropathy or renal vein thrombosis (or the need for dialysis in CAPS), cardiology for cardiac valvular lesions or pulmonary hypertension, and obstetrics/gynecology for pregnant teens or counseling on contraception and puberty. Even within rheumatology, APS may benefit from combined adult and pediatric rheumatology input as the patient transitions to care in adulthood.
Close coordination is also essential for discharge planning, including arranging outpatient anticoagulation monitoring (many pediatric centers have anticoagulation clinic nurses or pharmacists), obtaining insurance approval for medications such as enoxaparin or warfarin, and educating the family. Support from social work and pharmacy is valuable to ensure access to and compliance with therapy.
Prognosis and Long-Term Follow-Up
While pediatric APS is chronic and can significantly impact life, many children can thrive with appropriate management. Recurrent thrombosis is a significant concern; in one series, nearly 43% of pediatric APS patients experienced recurrent clots over a ~6-year follow-up, often linked to subtherapeutic anticoagulation or non-adherence. This highlights the necessity for strict adherence to anticoagulation protocols and regular monitoring. With proper anticoagulation, the recurrence rate can be minimized.
Long-term anticoagulation is generally recommended for several years and often indefinitely, especially if the child has “high-risk” APS (arterial thrombosis, multiple events, or triple-positive antibodies). If a decision is made to stop warfarin after a provoked single event, it should be done in collaboration with specialists and with very close follow-up; generally, the antiphospholipid antibodies should be rechecked. If they remain strongly positive, most would continue anticoagulation indefinitely.
Children with APS require regular follow-ups with rheumatology (or hematology) to monitor for new symptoms and manage their treatment. For secondary APS, controlling the primary disease (e.g., maintaining SLE in remission) is crucial to reduce further damage—active lupus can increase the risk of thrombosis. Over time, chronic damage can accumulate; for example, some may develop chronic deep vein insufficiency from DVTs or cognitive effects from strokes. The Damage Index for Thrombotic Antiphospholipid Syndrome (DIAPS) has been used in research to quantify organ damage in APS patients; even pediatric patients can accrue damage over time, underscoring the importance of prevention and early intervention.
Transitioning to adult care is a pivotal aspect of managing pediatric APS. As these patients reach adulthood, they must transition to adult rheumatologists or hematologists who will continue their care. This transition should be carefully planned to ensure that the new providers are well-informed about the patient’s history. Adolescent patients should be gradually educated to take ownership of their disease management (for instance, learning how to manage their anticoagulation, understanding the interaction of warfarin with diet/medications, etc.).
Prognosis in pediatric APS varies: those with secondary APS (lupus) may experience more ongoing disease activity and organ involvement (thus potentially resulting in more complications), whereas a child with primary APS whose thromboses are well-managed might enjoy long asymptomatic periods. The mortality rate of pediatric APS is not well defined due to its rarity. Still, it is undoubtedly lower than that seen in historical adult APS cohorts, except in cases of catastrophic APS. Catastrophic APS is associated with a high mortality rate (~30–50% even with therapy in adults); pediatric data are limited, but it is a severe condition that requires aggressive treatment.
In summary, pediatric APS is a chronic condition that requires long-term follow-up for anticoagulation management, monitoring of antibody status (though aPL often remain positive indefinitely), and screening for new manifestations (e.g., a child with APS should have periodic checks for hypertension, proteinuria, etc., as aPL can cause renal microvascular damage even without obvious clinical events). With vigilant care, children with APS can lead active lives, but they will need ongoing precautions and collaboration between pediatric and adult specialists over time.
Conclusion
TMA and APLA in children present challenging, high-stakes diagnoses that require timely recognition and a coordinated, evidence-based approach. Pediatric TMAs are most often associated with HUS syndromes, necessitating supportive care and sometimes specialized therapies (plasma exchange or complement inhibition). In contrast, pediatric APS is a rare autoimmune coagulopathy that requires anticoagulation and often immunomodulation. Front-line inpatient providers should maintain a high index of suspicion for these conditions in critically ill children with thrombocytopenia, hemolysis, or thrombosis and initiate appropriate workup and management promptly. Early involvement of pediatric subspecialists, including nephrology, hematology, and rheumatology, among others, is key to optimizing outcomes. With advances such as eculizumab for aHUS and improved protocols for TTP and APS, the outlook for affected children continues to improve, but long-term follow-up and multidisciplinary care remain essential for managing these complex syndromes and their sequelae.
References and further reading:
1. Loirat C. et al. (2016) – Pediatric Nephrology.
International Consensus on Managing Atypical HUS in Children.
- Summary: A global panel of pediatric nephrologists has established guidelines for diagnosing and treating atypical hemolytic uremic syndrome (aHUS) in children. It emphasizes the importance of promptly ruling out other thrombotic microangiopathies (like TTP via ADAMTS13 testing and STEC-HUS) and initiating complement inhibition therapy early. The consensus reflects the dramatic improvement in outcomes with eculizumab, the first effective complement-blocking drug, which has “dramatically changed the outlook” of this once often-fatal disease.
- Key Clinical Takeaway: Immediate workup and treatment are critical – test for ADAMTS13 activity and Shiga toxin promptly to distinguish aHUS from TTP or typical HUS, then initiate eculizumab without delay if aHUS is suspected. Plasma exchange, which was once considered a first-line treatment, is now regarded as a second-line approach to targeted therapy. The guideline also poses unanswered questions (e.g., optimal duration of treatment) and calls for ongoing research.
- Relevance: Inpatient pediatricians and rheumatologists encounter TMA cases where rapid differentiation is crucial for lifesaving outcomes. This consensus provides a diagnostic algorithm and treatment roadmap for pediatric aHUS, underscoring that early complement blockade can be organ- and life-saving. It guides hospitalists in coordinating urgent diagnostics (ADAMTS13 assays, infectious studies) and initiating eculizumab, in consultation with specialists, to improve outcomes in children with aHUS.
2. Greenbaum L.A. et al. (2016) – Kidney International.
Pivotal trial of eculizumab in pediatric aHUS.
- Summary: This open-label Phase II study evaluated weight-based eculizumab in twenty-two children (5 months to 17 years) with atypical HUS. After 26 weeks, most displayed a complete thrombotic microangiopathy response, with normalized blood counts and improved renal function. Notably, 9 of 11 children on dialysis at baseline were able to discontinue dialysis after eculizumab therapy. No new safety concerns emerged (no meningococcal infections or deaths).
- Key Clinical Takeaway: Eculizumab has proven to be highly effective and safe in children with aHUS, inducing hematologic remission and improving or preserving kidney function in the majority of cases. The study supports the practice of early, upfront eculizumab in pediatric aHUS, rather than plasma exchange. The authors conclude that immediate initiation of eculizumab upon diagnosis is warranted in children, given the substantial benefits.
- Relevance: This landmark trial underpins current inpatient practice for pediatric aHUS. It provides hospital providers with confidence that eculizumab can reverse TMA and avert end-stage renal failure in children. Pediatric rheumatologists and nephrologists utilize this evidence to advocate for timely access to complement inhibition in hospitalized children with aHUS, and it informs monitoring (vaccination for meningococcus and signs of improvement) during therapy.
3. Walsh P.R. & Johnson S. (2018) – Archives of Disease in Childhood.
Review of HUS in children (STEC-HUS, aHUS, and others).
- Summary: This high-yield review outlines the three primary forms of hemolytic uremic syndrome in pediatrics: typical Shiga toxin–mediated HUS, atypical complement-mediated HUS, and Streptococcus pneumoniae-associated HUS. It discusses the latest management strategies for each and traces how the field has transformed with the discovery of complement’s role in aHUS and the “dawn” of complement inhibition therapy. The authors provide practical guidance from the prodrome (bloody diarrhea) through fulminant HUS, including when to initiate plasma exchange, dialysis, or eculizumab.
- Key Clinical Takeaway: Supportive care (volume management, blood pressure control, dialysis if needed) remains the cornerstone for typical STEC-HUS, as no specific anti-toxin therapy has proven effective. Antibiotics are generally avoided during the diarrhea phase due to the potential to worsen toxin release. In contrast, for atypical HUS, the article emphasizes that eculizumab is the standard of care, having significantly improved outcomes in aHUS. It also highlights pneumococcal HUS, recommending antibiotic treatment of the invasive infection and noting the need for complement-directed therapy in severe cases.
- Relevance: Inpatient pediatricians frequently manage children with HUS following diarrheal illness; this review provides precise management algorithms and flags warning signs of atypical HUS. Pediatric rheumatologists benefit from the discussion of complement-mediated HUS, as they may co-manage these patients. Overall, this paper equips clinicians with practical, up-to-date approaches for HUS on the wards, from intensive care support to deciding when to involve subspecialists for plasma exchange or eculizumab.
4. Tanaka K. et al. (2021) – Pediatric Nephrology.
Ravulizumab (long-acting C5 inhibitor) in pediatric aHUS.
- Summary: This study examined 10 children with aHUS who were switched from chronic eculizumab to ravulizumab, a longer-acting C5 complement inhibitor. Dosing every 4–8 weeks (instead of every 2 weeks with eculizumab) maintained disease remission. Over 52 weeks, kidney function remained stable or improved, and hematologic parameters remained normal. No patient required new dialysis, and no new safety issues (e.g., no meningococcal infections) emerged.
- Key Clinical Takeaway: Ravulizumab is efficacious and safe in children with aHUS, providing sustained TMA control with a much more convenient dosing interval. This translates to fewer infusions (every 1–2 months vs. biweekly), which can reduce the treatment burden on families while maintaining the same level of disease control. All patients in the trial remained at or below their baseline chronic kidney disease stage throughout the study.
- Relevance: For inpatient teams and rheumatology/nephrology specialists, this development means that children on complement inhibition can be transitioned to a more patient-friendly regimen without compromising safety or efficacy. Hospitals may see fewer infusion visits and better adherence. When initiating complement blockade in a new aHUS case or maintaining a long-term therapy plan, ravulizumab offers an appealing option that can enhance the quality of life for pediatric patients while ensuring ongoing TMA suppression.
5. Taylor A. et al. (2024) – Blood Advances.
Caplacizumab in pediatric TTP (UK registry study).
- Summary: This multi-center UK study reported on 16 children and adolescents with immune TTP who received caplacizumab (an anti-von Willebrand factor agent) alongside standard plasma exchange and immunosuppression. Four patients were under 12 years old, including one toddler. Caplacizumab was administered daily, with a lower dose for the youngest children, and achieved rapid platelet count recovery (median 5.5 days) in all cases. Disease control was excellent: no patients experienced refractory TTP, and only one relapse occurred during the follow-up. One child had a severe nosebleed that was managed by temporarily holding caplacizumab, but there were no life-threatening bleeds.
- Key Clinical Takeaway: Caplacizumab is a game-changer for TTP, and this study demonstrates its effectiveness and apparent safety in pediatric patients as well. Children treated with this approach experienced a faster resolution of TTP episodes and required fewer plasma exchange sessions compared to historical experiences, mirroring the results of adult trials. The drug’s main side effect—bleeding—was manageable. This supports the use of caplacizumab in children ≥40 kg (approved in adolescents) and even off-label in smaller kids with severe TTP, in conjunction with plasma exchange and steroids, to improve outcomes.
- Relevance: Pediatric TTP is a rare but life-threatening condition; this study provides reassurance to hospital providers that caplacizumab can be used in children to control TTP and reduce ICU stays. For pediatric rheumatologists managing TTP secondary to lupus or other autoimmune diseases, caplacizumab offers targeted therapy to halt the microthrombotic process. In practice, this means inpatient teams can achieve quicker platelet normalization and possibly reduce plasma exchange days, which is “particularly relevant in pediatric care” where lengthy ICU procedures have significant impacts.
6. Wincup C. & Ioannou Y. (2018) – Frontiers in Pediatrics.
Differences between childhood-onset and adult APS.
- Summary: This review compares pediatric antiphospholipid syndrome (APS) with adult APS in terms of epidemiology, clinical features, and management. Unlike the female-predominant adult APS, childhood APS has a more balanced gender distribution. Children are more likely to have APS secondary to lupus and often present with thromboses in the CNS (strokes) or hematologic manifestations (thrombocytopenia, hemolytic anemia) rather than the obstetric complications seen in adults. Notably, children with APS face a higher risk of recurrent thromboembolism than adults. The authors highlight the lack of pediatric-specific trials, indicating that treatment in children is primarily based on adult protocols. Long-term anticoagulation is usually recommended after a first thrombosis, but this must be balanced with lifestyle considerations (e.g., restrictions on contact sports).
- Key Clinical Takeaway: Pediatric APS is not merely “adult APS in small patients.” It has distinctive features – for instance, a child with APS is more likely to have an underlying lupus and to experience non-criteria manifestations like migraine, chorea, or livedo. Management should be proactive in preventing recurrent clots (often indefinite anticoagulation following any serious thrombosis). Still, providers must customize therapy to meet a growing child’s needs (monitoring warfarin in a teenager can be challenging, and using low-molecular-weight heparin or newer anticoagulants may be considered on a case-by-case basis). Adjunct treatments, such as low-dose aspirin or hydroxychloroquine, which are standard in adult APS with SLE, show potential benefits in children but lack pediatric trial evidence.
- Relevance: This article serves as a valuable guide for pediatric rheumatologists and hospitalists, emphasizing that criteria and treatments derived from adults may not fully apply to children. It encourages a heightened awareness of APS in pediatrics (for example, a teenage boy with a stroke may have APS despite the condition being rarer in males) and highlights the importance of multidisciplinary management. For inpatient care, it emphasizes the importance of meticulous anticoagulation management in children and vigilance for recurrent thrombosis in pediatric APS patients.
7. Nageswara Rao A. et al. (2017) – Blood Coagulation & Fibrinolysis.
Mayo Clinic series on pediatric APS outcomes.
- Summary: This 20-year retrospective review from a tertiary center described 17 children diagnosed with APS (median age ~15 years at first thrombosis). Just over half had primary APS (no systemic disease), while the rest were associated with lupus or other conditions. Over a median follow-up of ~4 years, 59% experienced recurrent or progressive thromboses, which is considerably higher than typically reported in adults. Most recurrences were venous clots, and importantly, 80% of those children were not therapeutically anticoagulated at the time of recurrence. Some events occurred more than a year after the initial clot, indicating a long-term risk.
- Key Clinical Takeaway: Recurrent thrombosis is a common occurrence in pediatric APS, often attributed to suboptimal anticoagulation. This study’s stark finding—that nearly three in five children had another clot—suggests that indefinite, well-managed anticoagulation is likely indicated after a first APS thrombotic event in children. Many recurrences happened when warfarin levels were not therapeutic or therapy had been stopped, underscoring the need for better adherence support and closer monitoring for pediatric patients. The authors call for larger studies but lean toward recommending long-term (even lifelong) anticoagulation in pediatric APS, given the high recurrence rate when anticoagulation was shortened.
- Relevance: Inpatient pediatricians may be the first to diagnose APS in a child with a new thrombosis. These findings inform discharge planning, highlighting that a short course of anticoagulation (3–6 months) is usually insufficient in APS. Pediatric rheumatologists will use this evidence to justify prolonged anticoagulation to families, despite the inconvenience, because the risk of a second clot (stroke, PE, etc.) in childhood APS is very high. This study ultimately shifts practice toward a mindset of APS = long-term anticoagulation in kids, similar to adult APS management, but with even greater vigilance.
8. Ma J. et al. (2018) – Clinical Rheumatology.
Analysis of 58 pediatric APS patients (multi-center).
- Summary: This larger cohort study (n = 58 children) provides a comprehensive view of pediatric APS. The mean age was approximately 14 years; 76% of the cases were secondary to systemic lupus erythematosus, while only 24% were primary, indicating that pediatric APS often accompanies SLE. Venous clots, particularly deep vein thromboses of the legs, were the most common events (about 37% of patients), followed by strokes and other arterial thrombi. More than half the children exhibited the complete “triple-positive” antibody profile (lupus anticoagulant, anticardiolipin, and anti-β2GPI), and almost all (95%) tested positive for lupus anticoagulant. During a median follow-up of approximately 3 years, 13% of patients experienced recurrent thrombosis; notably, all recurrences occurred among those who tested double- or triple-positive for antibodies. The authors found no significant difference in initial thrombosis rates between single- and triple-antibody-positive patients; however, multi-positive patients were clearly at an increased risk for subsequent events. They recommend prolonged anticoagulation (or antiplatelet therapy) for children with high-risk antibody profiles to prevent recurrence.
- Key Clinical Takeaway: In pediatric APS, the presence of multiple aPL antibodies (especially lupus anticoagulant) indicates a higher risk and should inform management. Children with triple-positive APS should be considered for long-term anticoagulation to prevent recurrence, even after their first event, given this study’s observation that all repeat clots were found in the triple-positive group. Additionally, the prevalence of lupus-associated APS in this cohort reminds clinicians to routinely check aPL antibodies in any pediatric lupus patient – about 1 in 5 of these children may develop APS-related clots. Conversely, a child with “primary” (non-lupus) APS requires close monitoring for future autoimmune disease.
- Relevance: This study provides pediatric rheumatologists with data to stratify risk. A teenager with SLE, nephrotic-range proteinuria, and triple-positive aPL requires aggressive prophylaxis and follow-up. In contrast, a child with a single-positive antibody may be managed with standard anticoagulation duration plus risk factor management. For inpatient doctors, it emphasizes that when a child is admitted with thrombosis and found to have aPL, screening for underlying SLE is essential (since two-thirds of pediatric APS patients have lupus). It also supports collaborative decision-making regarding extended anticoagulation in high-risk pediatric antiphospholipid syndrome (APS) cases.
9. Madison J. et al. (2022) – Pediatric Rheumatology.
Contemporary single-center experience with pediatric APS.
- Summary: This retrospective study of 21 APS patients at a large children’s hospital offers insights into the disease spectrum and damage accumulation. The median age at APS diagnosis was 16, and slightly more patients had secondary APS (mostly lupus) than primary. Notably, two-thirds of the children exhibited “non-criteria” APS manifestations – features such as thrombocytopenia, Coombs-positive hemolytic anemia, or livedo racemosa that are not included in the formal classification criteria. These occurred alongside classic thromboses. Almost half the cohort experienced at least one recurrent thrombosis (often due to subtherapeutic or interrupted anticoagulation, echoing other studies). The authors also evaluated organ damage using the APS Damage Index and found significant chronic morbidity in both primary and secondary APS cases. They emphasize the need for pediatric-specific APS criteria, given that children frequently exhibit features not captured by adult criteria.
- Key Clinical Takeaway: Recognize the broader APS phenotype in children. A child with persistent aPL may present with multisystem features like cytopenias, skin livedo, or migraine, even before an extensive vessel thrombosis occurs. These “red flags” should prompt consideration of APS and the implementation of preventive measures. This study reinforces that adherence to anticoagulation is critical: many recurrences happened when teens were non-adherent or subtherapeutic on warfarin. It also suggests that pediatric rheumatologists should utilize tools like DIAPS (Damage Index in Thrombotic APS) to monitor cumulative organ damage in these patients.
- Relevance: For hospitalists, this means that a child admitted with, for example, hemolytic anemia and low platelets in the context of lupus might not just be experiencing a lupus flare – it could be an APS-related microangiopathy, even if no major clot is visible. Early rheumatology input is warranted in such cases. Pediatric rheumatologists should recognize that they often need to extend beyond adult APS criteria to diagnose and manage children effectively. For instance, treating a high-risk aPL-positive child prophylactically, even without criteria-level thrombosis, may be reasonable. Overall, this study urges clinicians to think holistically about APS in kids and to strive for strategies (education, possibly novel anticoagulants or adherence aids) to prevent adolescents with APS from lapsing in medication adherence, which directly ties to inpatient recurrences.
10. Go E.J.L. & O’Neil K.M. (2017) – Current Opinion in Rheumatology.
Catastrophic antiphospholipid syndrome (CAPS) in children.
- Summary: This review focuses on the most severe and acute presentation of APS—catastrophic APS—characterized by rapid and diffuse microvascular thromboses, which can lead to multi-organ failure. Although CAPS is rare in pediatrics, it carries a mortality rate of 33–50% and often affects children with active lupus or infections. The authors note that infections are a common trigger in pediatric CAPS and that complement activation plays a significant role in the pathogenesis of the fulminant thromboses. Treatment must be aggressive and multimodal, including anticoagulation, high-dose steroids, plasmapheresis, and/or IVIG, as well as immunosuppression (e.g., cyclophosphamide or rituximab), to halt autoantibody production. Importantly, emerging evidence suggests that complement inhibitors, such as eculizumab, can improve outcomes in CAPS by mitigating the thrombotic storm. The review emphasizes the importance of prompt recognition and the necessity of eliminating any infection that triggers the cascade.
- Key Clinical Takeaway: CAPS is a pediatric rheumatologic emergency. It demands simultaneous therapies: initiate heparin, broad-spectrum antimicrobials, and high-dose steroids at the first suspicion, and involve specialists for plasmapheresis and IVIG. Given the data on complement, many experts now include eculizumab in the treatment of refractory or severe pediatric CAPS, especially if ADAMTS13 is normal (ruling out TTP) and there is evidence pointing to APS. Early use of eculizumab, combined with therapies such as rituximab to suppress antiphospholipid antibody generation, may tip the balance in favor of survival in what is otherwise often a fatal condition.
- Relevance: Hospital teams in the ICU must recognize CAPS in children with disseminated thromboses, which often requires suspicion in a child with SLE or APS who suddenly develops multi-organ failure with TMA findings. This article equips clinicians with knowledge of cutting-edge interventions (e.g., complement blockade) to rescue such patients. For pediatric rheumatologists, the review consolidates current practice, recommending a “triple therapy” approach (anticoagulation, corticosteroids, and plasmapheresis/IVIG) plus targeted additions (infection control, eculizumab, and immunosuppressants) as the preferred treatment. Being aware of CAPS is crucial for inpatient providers, as prompt and aggressive treatment is key to improving the high mortality associated with this syndrome.