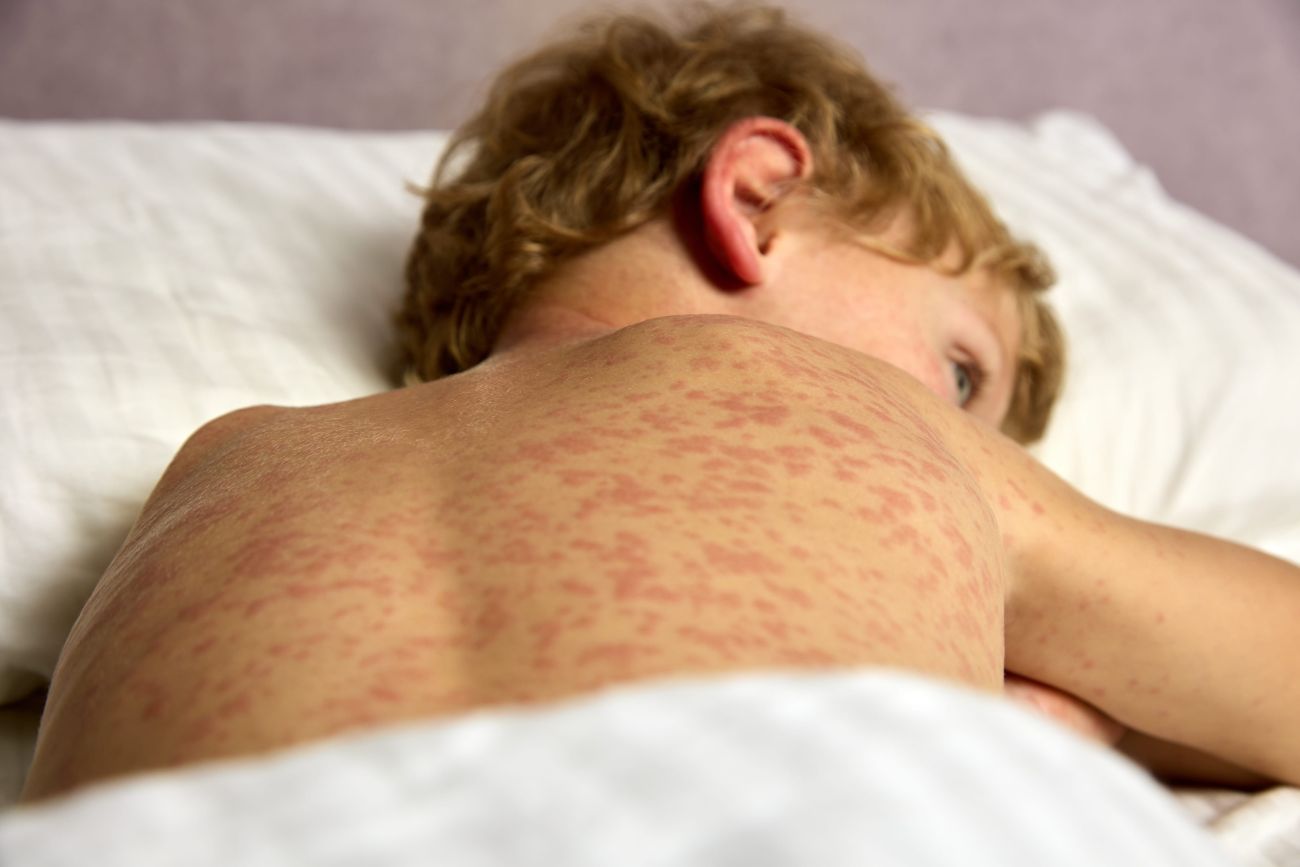
Background and Rationale for Reclassification
Systemic juvenile idiopathic arthritis (sJIA), formerly known as Still’s disease in children, was traditionally classified as a subtype of JIA. However, increasing evidence shows that sJIA and adult-onset Still’s disease (AOSD) are essentially the same disorder occurring at different ages. The arbitrary age cutoff of 16 years, which separated “juvenile” from “adult” forms, is now seen as an artificial distinction. A 2023 EULAR/PReS task force led by De Benedetti, Fautrel, and colleagues reviewed clinical and laboratory data and found no major differences between pediatric sJIA and adult Still’s disease, apart from age-related reporting biases (e.g., adults more often report sore throat or myalgias). Both exhibit hallmark features like quotidian fevers, evanescent rash, arthritis or arthralgia, and hyperinflammation, with remarkably similar gene expression profiles and cytokine patterns in children and adults. Based on this strong overlap, experts concluded that these conditions represent one disease, “Still’s disease,” rather than two separate entities. Unlike other JIA subtypes, which are mainly autoimmune, Still’s disease is recognized as a distinct autoinflammatory syndrome driven by the innate immune system. It lacks the typical autoantibodies seen in autoimmune arthritis and instead involves dysregulated inflammasome-related cytokines, notably IL-1β and IL-18, along with neutrophil and macrophage activation. This autoinflammatory mechanism aligns Still’s disease more closely with periodic fever syndromes than with classic polyarticular JIA. Based on pathophysiology and clinical evidence, the task force recommended reclassifying sJIA as Still’s disease—removing it from the JIA category—and unifying pediatric and adult cases under a single name and diagnostic framework. The goal is to enhance recognition and treatment by acknowledging Still’s disease as a distinct systemic inflammatory disorder rather than a subtype of arthritis.
Terminology and Classification Changes
Under the new consensus, “Still’s disease” is the recommended umbrella term for this condition at any age. The term honors Sir George Still, who first described the childhood disease in 1897, and emphasizes the continuum between sJIA and AOSD. International experts agreed above 90% in favor of adopting a single name. Consequently, sJIA is no longer viewed just as a JIA subset but as the pediatric-onset form of Still’s disease, a systemic autoinflammatory disease. This change in terminology is supported by the Pediatric Rheumatology International Trials Organization (PRINTO) and other groups proposing new classification criteria. Notably, PRINTO’s 2019 classification proposal suggested that systemic JIA be removed from the JIA category and reclassified as an autoinflammatory disease based on its unique pathobiology. EULAR/PReS also highlight that future criteria should be unified across all ages—possibly using or adapting existing adult criteria, such as Yamaguchi’s, for all patients. In fact, the widely used Yamaguchi criteria for AOSD, which require fever, rash, and other symptoms, and exclude infections or autoimmune causes, have been validated in children with high sensitivity. Importantly, unlike older ILAR JIA criteria, Yamaguchi’s do not require arthritis to be present for diagnosis, aligning with the current understanding of Still’s disease onset. The task force explicitly recommends that persistent arthritis should no longer be mandatory to classify Still’s disease. Many children initially experience fever and systemic inflammation with only arthralgia, and arthritis may develop weeks or months later. Therefore, insisting on arthritis for diagnosis can cause harmful delays. In summary, the international consensus now classifies Still’s disease as a distinct entity—which includes sJIA and AOSD—within the spectrum of autoinflammatory syndromes rather than as a subset of autoimmune arthritis. This change in terminology and classification aims to promote earlier recognition, appropriate therapies, and integrated research in both children and adults.
Pathophysiology: An Autoinflammatory Disorder
Still’s disease is characterized by a dysregulated innate immune response with massive cytokine release. Unlike typical rheumatoid arthritis, adaptive immunity (T/B cells, autoantibodies) is not the main driver. Instead, macrophages and neutrophils play a central role, producing high levels of interleukin-1 (IL-1), IL-6, IL-18, and S100-family calgranulins, among other mediators. This cytokine environment causes the clinical features of daily spiking fevers, rash, and systemic inflammation—essentially a chronic autoinflammatory fever syndrome. IL-1β and IL-18 are both downstream of inflammasome activation, implicating innate immune complexes in the disease process. Notably, IL-18 is profoundly elevated in active Still’s disease (often 10-100 times normal), a unique feature not seen in most other rheumatic diseases. IL-18, along with S100A8/A9 (calprotectin) and S100A12, serves as a biomarker of innate immune activation and correlates with disease activity. Similarly, ferritin levels are usually very high (often thousands of ng/mL), reflecting macrophage activation; a glycosylated ferritin fraction below 20% is characteristic of Still’s disease in adults. These biomarkers—ferritin (especially hyperferritinemia), S100 proteins, and IL-18—have demonstrated similar diagnostic utility in both pediatric and adult populations. In fact, a systematic review identified these three as the most reliable biomarkers for Still’s disease across all ages. Genetic studies also support a common underlying pathogenesis: certain HLA associations and cytokine gene polymorphisms (e.g., variants affecting IL-1 or IL-6 pathways) are shared between sJIA and AOSD. Unlike other forms of JIA, Still’s disease does not associate with autoantibodies (rheumatoid factor and ANA are typically negative), reinforcing that it is not a classic autoimmune disorder. Instead, it behaves as a complex polygenic autoinflammatory syndrome driven by innate immunity. Over time, some patients with chronic arthritis may develop features of adaptive immunity (such as joint erosions or even ANA positivity in a minority), but the primary and central pathology remains an innate immune “storm.” This is exemplified by the disease’s most severe complication, macrophage activation syndrome (MAS), which is essentially a form of secondary hemophagocytic lymphohistiocytosis (HLH) caused by excessive innate immune activation. In Still’s disease, MAS occurs in up to 10-15% of patients, reflecting the tendency for uncontrolled cytokine cascades (IL-1, IL-18, IFN-γ) if the disease is not properly managed. Overall, the pathophysiology of Still’s disease is now understood to resemble systemic autoinflammatory diseases more than autoimmune arthritis, explaining its dramatic response to cytokine-targeted therapies.
Clinical Features and Updated Diagnostic Criteria
Clinically, Still’s disease presents with a characteristic constellation of systemic inflammatory features. The EULAR/PReS task force has proposed operational diagnostic criteria to recognize Still’s disease across all age groups (pediatric and adult).
Key features include:
1. Quotidian Fever: Daily spiking fevers ≥39°C that persist for at least 7 days. Fevers often spike once or twice daily (typically in the late afternoon or evening) with rapid defervescence in between.
2. Evanescent Rash: A transient salmon-pink rash, often accompanying fever spikes. The rash usually involves the trunk and proximal limbs and may be fleeting (evanescent), appearing with high fever and fading as temperature normalizes. It is classically non-pruritic and macular; in some cases, a urticarial or polymorphic rash can occur and still be consistent with Still’s.
3. Arthralgia/Arthritis: Musculoskeletal involvement is common, typically manifesting as arthralgias or myalgias early in the disease. Overt arthritis may develop later (often weeks into the illness), and while it supports the diagnosis, arthritis is not required at disease onset. Especially in children, arthritis may only appear after a period of systemic symptoms (median ~1 month delay). Thus, a patient can be diagnosed with Still’s disease based on systemic features even before arthritis emerges, to avoid delays in care. (Other JIA subtypes, in contrast, require chronic arthritis for diagnosis; this is a key change in Still’s disease criteria.)
4. Marked Inflammation: Evidence of hyperinflammation is present, with neutrophil-predominant leukocytosis and highly elevated acute phase reactants. Common laboratory findings include very high C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), elevated ferritin, and often thrombocytosis. Ferritin levels are frequently markedly increased (e.g., >1000 ng/mL), and a rising ferritin in the context of persistent fever and cytopenias should prompt evaluation for impending MAS. Mild liver enzyme elevations are also common.
By definition, other causes, such as infections and malignancies, must be excluded, as Still’s disease is a diagnosis of exclusion. These criteria align with the classical descriptions of Still’s disease and are consistent with existing classification frameworks (e.g., Yamaguchi’s criteria for AOSD and the ILAR/PRINTO criteria for sJIA). Notably, the 2019 PRINTO classification for sJIA (Martini et al.) already allowed for a diagnosis based on fever and systemic features, even in the absence of immediate arthritis, similar to the adult criteria. The new consensus affirms this approach: requiring 6 weeks of arthritis (as per older ILAR criteria) is no longer necessary to classify Still’s disease. In practice, a clinician should strongly suspect Still’s disease in any child or adult with unexplained daily fevers of at least a week’s duration, the typical rash, and elevated inflammatory markers (especially if ferritin or IL-18 is very high), after ruling out infections, malignancy, and other inflammatory diseases. Traditional adult Still’s criteria (Yamaguchi or Fautrel) can be applied to children and vice versa to aid diagnosis. Supporting features often seen in Still’s disease include lymphadenopathy, hepatosplenomegaly, and serositis (e.g., pericarditis), which were part of earlier criteria and remain common in severe cases. Another clue is the absence of autoantibodies: Still’s patients are typically RF-negative and ANA-negative, which helps distinguish them from autoimmune forms of juvenile or adult arthritis. Additionally, extremely high serum ferritin or IL-18 can serve as useful ancillary markers pointing toward Still’s. Overall, the updated diagnostic approach emphasizes early recognition of the core clinical syndrome of Still’s disease (fever, rash, systemic inflammation) even before chronic arthritis is evident, enabling prompt therapy to prevent complications.
Key Biomarkers in Still’s Disease
As an autoinflammatory condition, Still’s disease is associated with distinctive biomarker profiles that reflect innate immune activation:
Serum Ferritin: Elevated ferritin levels are a hallmark; they often exceed 1000 ng/mL and can reach extremely high values. A ferritin >684 ng/mL is one criterion in the MAS classification, and in Still’s, it correlates with disease activity. Notably, a low glycosylated ferritin fraction (<20%) is often observed in adult Still’s disease (Fautrel criteria) and may also be seen in sJIA. Hyperferritinemia (disproportionate to CRP) in a febrile patient raises suspicion for Still’s or MAS.
S100 Proteins: S100A8/A9 and S100A12 (calgranulins) are markedly elevated in active Still’s. These neutrophil-derived damage-associated molecular patterns (DAMPs) are sensitive markers of innate immune activation. S100A8/A9 (also called calprotectin) levels correlate with sJIA activity and often rise before clinical flares, making it a potential predictive biomarker. Studies show that S100 proteins have diagnostic utility in differentiating Still’s disease from fever of unknown origin or other causes.
Interleukin-18: IL-18 is one of the most elevated cytokines in Still’s disease, with levels far exceeding those in other inflammatory conditions. It reflects macrophage inflammasome activation. Extremely high IL-18 (sometimes >10,000 pg/mL in serum) is highly specific to sJIA/AOSD and has been proposed as a diagnostic marker. High IL-18 levels also correlate with the risk of MAS, as IL-18 further drives the activation of NK and T cells. IL-18 binding protein (endogenous inhibitor) is often insufficient in Still’s, leading to excess free IL-18.
IL-6: IL-6 is elevated in active Still’s and contributes to fevers, rash, and acute phase response (IL-6 drives CRP production). IL-6 levels tend to normalize with effective therapy (e.g., tocilizumab).
Other Cytokines: IL-1β is elevated locally (e.g., in tissues) or can be inferred from gene expression, even if it is not always high in serum due to its short half-life. Interferon-γ may be elevated, especially during MAS. CXCL9 (an IFN-γ-inducible chemokine) is often elevated in MAS and can help indicate impending MAS in Still’s patients.
Routine Labs: Neutrophil count is typically high in most cases (often >10-15k with a neutrophil percentage>80%). Platelets are usually elevated during active disease (acute phase reactant). Liver enzymes can be mildly elevated. ESR/CRP are generally very high. If these markers are normal, Still’s is unlikely. During MAS, paradoxically, CRP may drop (because the liver shifts to producing fibrinogen and becomes overwhelmed), while ferritin skyrockets – this discordance is an essential clue to the onset of MAS.
Absence of Autoimmune Markers: As mentioned, RF and ANA are typically negative. Their presence should prompt consideration of an alternate or concomitant diagnosis.
These biomarkers are not disease-specific on their own, but when combined and considered in context, they support a diagnosis of Still’s disease. A systematic review confirmed that ferritin, S100A8/A9, and IL-18 are consistently the top biomarkers distinguishing Still’s disease, with similar diagnostic performance in both children and adults. Going forward, integrating biomarkers into classification and diagnostic criteria is an area of active research (for example, incorporating ferritin or IL-18 cut-offs to enhance specificity). For now, they serve as valuable tools for clinicians: for example, a very high IL-18 or calprotectin level in a child with fever strongly favors sJIA over infection, and a falling ferritin level can indicate a therapeutic response.
Treatment Implications of the Autoinflammatory Paradigm
The recognition of Still’s disease as an IL-1/IL-6–driven autoinflammatory condition has revolutionized its management. Traditional therapies like methotrexate or TNF inhibitors (effective in other JIA types) are less effective here. Instead, targeted inhibition of IL-1 and IL-6 has shown dramatic efficacy. Current first-line treatment recommendations for Still’s disease emphasize early use of IL-1 or IL-6 blockade:
Interleukin-1 inhibitors: IL-1 blockade can induce rapid remission of systemic symptoms. Anakinra (a recombinant IL-1 receptor antagonist) often leads to defervescence and improvement in rash and arthritis within days to weeks in sJIA/AOSD. High-dose daily anakinra is commonly used in children. Longer-acting IL-1 inhibitors, such as canakinumab (an anti–IL-1β monoclonal antibody) or rilonacept, can also be used. Studies and clinical experience show that IL-1 inhibition can achieve clinically inactive disease in a large proportion of patients, especially when started early.
Interleukin-6 inhibitors: IL-6 drives the intense inflammatory features (fever, acute phase reactants, anemia) of Still’s disease. Tocilizumab (anti–IL–6 receptor antibody) is highly effective for the systemic and arthritic manifestations of sJIA and AOSD. It is approved for sJIA in many countries and has demonstrated rapid normalization of fever and CRP, as well as improvement in arthritis. IL-6 blockade is an equally valid first-line biological option, particularly in patients who may not tolerate IL-1 inhibitors or have partial responses.
Head-to-head comparisons are lacking, but IL-1 and IL-6 inhibitors both demonstrate the highest levels of efficacy in Still’s disease, far superior to other immunomodulators. A systematic review of therapies found that the odds of achieving significant improvement (ACR50) were 6–8 times higher with IL-1 or IL-6 inhibitors than with placebo. By contrast, conventional DMARDs like methotrexate, or even TNF inhibitors, have only marginal benefits in pure Still’s disease. Therefore, modern guidelines strongly advocate starting an IL-1 or IL-6 inhibitor as soon as the diagnosis is confirmed rather than cycling through steroids and methotrexate as was done historically. This early introduction of biologics is aimed at quickly controlling the cytokine storm and preventing complications (like MAS or chronic arthritis).
Glucocorticoids (corticosteroids) are a double-edged sword in Still’s disease management. On the one hand, high-dose corticosteroids (e.g., IV methylprednisolone pulses or daily prednisone) are highly effective in quelling hyperinflammation and are lifesaving in acute MAS. Most patients with severe sJIA/AOSD will receive steroids initially. On the other hand, long-term steroid use causes significant toxicity (growth suppression, osteoporosis, Cushing’s syndrome, etc.), and with the advent of IL-1/IL-6 inhibitors, the goal is to minimize steroid exposure. The EULAR/PReS recommendations call for rapid tapering of steroids once biologics take effect. Ideally, patients should achieve clinically inactive disease on an IL-1/IL-6 inhibitor and wean off steroids within 3–6 months. This steroid-sparing approach is now standard, and many children have successfully avoided long-term prednisone by early use of anakinra or tocilizumab.
A treat-to-target (T2T) strategy is emphasized for Still’s disease, similar to other rheumatic diseases. The ultimate goal is complete remission off medication. To achieve this, interim targets are set: for example, the task force recommends that by approximately 1 month into therapy, fever should resolve and CRP levels normalize, and by 3 months, the patient should have clinically inactive disease on minimal or no steroids. If these targets are not met, escalation or a switch of therapy is advised (e.g., switching from IL-1 to IL-6 inhibitors or vice versa, or adding other agents). Once a patient maintains remission (inactive disease) for at least 3–6 months on biologic therapy, guidelines suggest considering tapering the biologic as well. In practice, many pediatric rheumatologists attempt to taper anakinra or tocilizumab after about 1 year of remission, with close monitoring for flares. A significant proportion of patients can eventually sustain drug-free remission, especially those treated early in the disease course. This marks a notable improvement from previous eras, when prolonged steroid dependence and chronic arthritis were common.
Other therapies are reserved for specific situations. Methotrexate may help with chronic arthritis in Still’s disease, but it does not effectively control systemic features and has a slow onset, so it is not used as a first-line standalone treatment. Cyclosporine is effective in MAS and has been used in sJIA with recurrent MAS or as a steroid-sparing agent in refractory cases. TNF inhibitors (like etanercept) were historically tried in sJIA, but response rates were low compared to IL-1/6 blockers. JAK inhibitors (e.g., baricitinib) are emerging as potential options; they can inhibit signaling of multiple cytokines, including IFN-γ and IL-6. Small studies suggest JAK inhibitors may help Still’s disease, especially refractory arthritis, but more evidence is needed. IVIG has been used anecdotally, particularly in MAS or when Kawasaki disease is considered, but it is not routine. Thalidomide and cyclophosphamide have been reported in refractory cases or those with lung complications, but their roles are not well established. Finally, experimental approaches targeting IL-18 (e.g., tadekinig alfa, an IL-18 binding protein) or IL-1β/IL-18 dual inhibition (such as a dual cytokine trap) are being studied to address the persistent elevation of these cytokines.
In summary, therapy for Still’s disease now focuses on blocking IL-1 and IL-6, rapidly controlling inflammation, and preventing long-term damage. This shift in approach, made possible by recognizing Still’s as an autoinflammatory cytokine-driven disease, has significantly improved outcomes. Many patients achieve remission and avoid the debilitating chronic courses previously observed past.
Management Guidelines and Consensus Recommendations
International guidelines have adapted to these advances. The 2023 EULAR/PReS consensus guidelines (published 2024) are the first unified recommendations for Still’s disease covering both children and adults. They present 4 main principles and 14 detailed recommendations related to diagnosis, treatment, and monitoring. Key points from these guidelines include:
- Unified Nomenclature: Reinforces that sJIA/AOSD are one disease, so all patients should be labeled “Still’s disease,” with age of onset noted as needed. This promotes a standardized approach across pediatric and adult rheumatology.
- Rapid Diagnosis: Clinicians should recognize the key features (fever, rash, arthralgia, lab markers) and exclude mimics to diagnose Still’s disease quickly. Waiting for chronic arthritis or damage is discouraged. The task force provided the operational criteria described above to help identify the disease early. They also recommend using established classification criteria (like Yamaguchi) flexibly for both age groups.
- Treatment-to-Target: Management should be based on shared decision-making with patients and families, aiming for clinically inactive disease (CID) and ultimately drug-free remission. Regular assessment of disease activity should guide adjustments to therapy. The task force defines CID as the absence of active joints, no fever or rash, normalized laboratory results, etc., and remission as maintaining CID for at least 6 months. Timeframes are suggested (e.g., improvement within 1 month, inactive by 3-6 months) to keep treatment on track. If targets are not met, consider changing biologics or adding therapy rather than continuing an ineffective regimen.
- First-Line Therapy: IL-1 or IL-6 inhibitors should be started as early as possible once Still’s disease is diagnosed. This is a strong recommendation (Level of Evidence 2b, with a Level of Agreement of approximately 96% among experts). Early biologic treatment is linked to higher remission rates. Conventional agents (methotrexate, etc.) are reserved for adjunctive use or milder cases.
- Glucocorticoid Use: Steroids are effective but carry significant risks. Guidelines advise using the lowest effective dose for the shortest time. A brief initial course of high-dose prednisone may control severe symptoms, but the goal is to taper off quickly (within 3–6 months) once IL-1/IL-6 inhibitors are effective. Prolonged steroid use should be avoided.
- Monitoring and Tapering: After remission is achieved on therapy, maintain it for at least 3–6 months before tapering the biologic. Tapering should be gradual and carefully monitored, as some patients may flare when biologics are withdrawn. The task force suggests that if remission is sustained, tapering off medication is a realistic goal for many, aligning with the treat-to-target approach of eventual drug-free remission.
- Macrophage Activation Syndrome: Due to the life-threatening nature of MAS, the guidelines emphasize prompt recognition and treatment. Physicians should have a high suspicion for MAS in any Still’s patient who deteriorates suddenly with persistent fever, worsening hyperferritinemia, cytopenias, rising liver enzymes, coagulopathy, or organomegaly. The 2016 MAS classification criteria (which include ferritin >684 ng/mL, platelet count <181k, AST >48, triglycerides >156 mg/dL, fibrinogen <250 mg/dL, etc., in a febrile sJIA patient) can assist in diagnosis. Once MAS is suspected, treatment must be immediate and aggressive. High-dose glucocorticoids are the first-line therapy for MAS (often IV pulse methylprednisolone). Additionally, anakinra (IL-1 inhibition) should be initiated or intensified, as IL-1 blockade has demonstrated efficacy in MAS. Many MAS treatment regimens also include cyclosporine A, which suppresses T-cell and macrophage activation and is especially recommended if there is an inadequate response to steroids alone. In refractory cases or severe pediatric MAS, intravenous immunoglobulin (IVIG) and etoposide (per HLH protocols) have been used. A newer option involves interferon-γ neutralization: the monoclonal antibody emapalumab (approved for primary HLH) has shown benefits in refractory MAS and is recommended in the guidelines as part of initial therapy for severe MAS alongside steroids and IL-1 inhibition. Early detection and combination therapy have significantly improved MAS outcomes.
- Pulmonary Involvement: The task force recognizes an “emerging issue” of Still’s disease-related lung disease in some patients. Rarely, children with long-standing or severe sJIA develop chronic interstitial lung disease or pulmonary alveolar proteinosis, especially in the context of prolonged high systemic inflammation. Because this complication can be subtle yet potentially fatal, active screening for lung disease is advised. Clinicians should monitor for signs such as digital clubbing, chronic cough, tachypnea, or hypoxia in Still’s patients. Baseline and periodic pulmonary function tests (like DLCO) may be considered in older children and adults, although in young children these are often not feasible. The guidelines recommend a low threshold for high-resolution chest CT if there are clinical concerns about lung disease. Early radiographic changes (e.g., ground-glass opacities) might prompt therapy adjustments. Interestingly, the consensus found no evidence that IL-1 or IL-6 inhibitors cause lung issues; therefore, the presence of “Still’s lung” does not contraindicate the continued use of IL-1/IL-6 blockers. Some cases of lung disease have been linked to high-dose corticosteroids and uncontrolled disease, so managing Still’s disease with biologics might actually reduce the risk. If lung disease is present, the task force recommends consulting centers experienced in Still’s lung complications. Treatments such as T-cell targeting agents (e.g., abatacept or other immunosuppressants) have been empirically used in severe lung cases, though no definitive treatment has been established yet. Ongoing research is investigating the connection between the IL-18 pathway and lung pathology in Still’s.
- Referral to Specialists: Refractory or severe cases—especially those with recurrent MAS or lung involvement—should be managed in collaboration with specialty centers experienced in Still’s disease. Multidisciplinary care involving rheumatology, hematology, pulmonology, and other specialties may be necessary.
These EULAR/PReS recommendations largely align with other guidelines worldwide. The ACR (American College of Rheumatology) 2021 guidelines for sJIA also prioritized IL-1 and IL-6 inhibitors as first-line therapy over methotrexate or TNF inhibitors and emphasized tapering steroids early. The ACR guidelines were created before the formal nomenclature change, so they refer to “sJIA,” but in practice, their treatment algorithm mirrors the Still’s disease approach. One notable addition by EULAR/PReS was the inclusion of explicit treat-to-target timeframes, which the ACR had not specified. Otherwise, there is broad international consensus on management. Both guidelines highlight vigilant monitoring for MAS, prompt treatment adjustments, and the importance of involving family and patients in care decision-making.
In clinical practice, reclassifying sJIA as Still’s disease has practical implications: pediatric and adult rheumatologists now approach these patients with a unified perspective, making the transition of care from childhood to adulthood smoother. Research protocols and clinical trials can also be designed to include both children and adults with “Still’s disease,” increasing the available evidence. Drug approvals may expand—such as treatments labeled for AOSD being used in sJIA and vice versa—since regulators now recognize them as the same disease entity.
Conclusion
The evolving view of systemic JIA as Still’s disease—a prototypical autoinflammatory condition—represents a paradigm shift in rheumatology. This reclassification is supported by strong evidence: sJIA and adult Still’s share identical clinical features, biomarker profiles, and pathogenic mechanisms. By removing the constraints of the old JIA subtype definition, clinicians can diagnose Still’s disease based on its unique clinical syndrome of fever, rash, and inflammation, without waiting for chronic arthritis to manifest. The endorsement of the name “Still’s disease” by international organizations (EULAR/PReS) symbolizes this change. More importantly, it aligns the management of children and adults, ensuring patients receive prompt, targeted therapy to IL-1/IL-6 and aggressive care for complications. The impact on outcomes has been remarkable—many patients achieve remission, and mortality from complications like MAS has decreased with earlier intervention. Ongoing research is further clarifying the immunologic drivers (such as IL-18 and innate immune genes) and exploring new treatments (e.g., IL-18 inhibitors, JAK inhibitors) for Still’s disease. As our understanding deepens, the hope is to cure this disease at its root by precisely modulating the autoinflammatory pathways. For now, the consensus is clear: systemic JIA/AOSD is Still’s disease, a single disease entity characterized by autoinflammation, and should be approached with unified diagnostic criteria and modern cytokine-targeted therapies. This refined framework improves patient care across all ages and demonstrates how re-examining disease classification can directly lead to better clinical outcomes.
References:
Recent peer-reviewed sources and international guidelines have been utilized in the above review, including the 2023 EULAR/PReS consensus statement on Still’s disease, systematicreviews of clinical and biomarker data, and current evidence on treatment efficacy, as well as the 2021 ACR guidelines and other key literature on sJIA/AOSD. These reflect the most up-to-date understanding of Still’s disease classification, pathophysiology, and management.