Overview of Pediatric Vasculitis
Vasculitis refers to inflammation of the walls of blood vessels, which can lead to tissue ischemia and damage to multiple organs. In children, vasculitis syndromes range from relatively common, self-limited illnesses to rare, life-threatening diseases. The most common pediatric vasculitis is IgA vasculitis, formerly known as Henoch-Schönlein purpura (HSP), which affects children aged 2–8. The next most common is Kawasaki disease, a medium-vessel vasculitis and the leading cause of acquired heart disease in children in developed countries. These two conditions account for the majority of childhood vasculitis cases. Other forms (e.g., ANCA-associated vasculitis, polyarteritis nodosa, Takayasu arteritis, Behçet’s) are much rarer in pediatric management and follow-up strategies, which are cognized due to their severity.
Early recognition of vasculitis by general practitioners (GPs) is crucial. Some vasculitides have distinct presentations (“red flags”) that should not be overlooked, as timely treatment can prevent serious complications. Below, we outline key vasculitis syndromes in children, the red flags to watch for, initial workup, when to refer to specialty care, and how to manage and follow these patients in the outpatient setting.
Henoch-Schönlein Purpura (IgA Vasculitis)
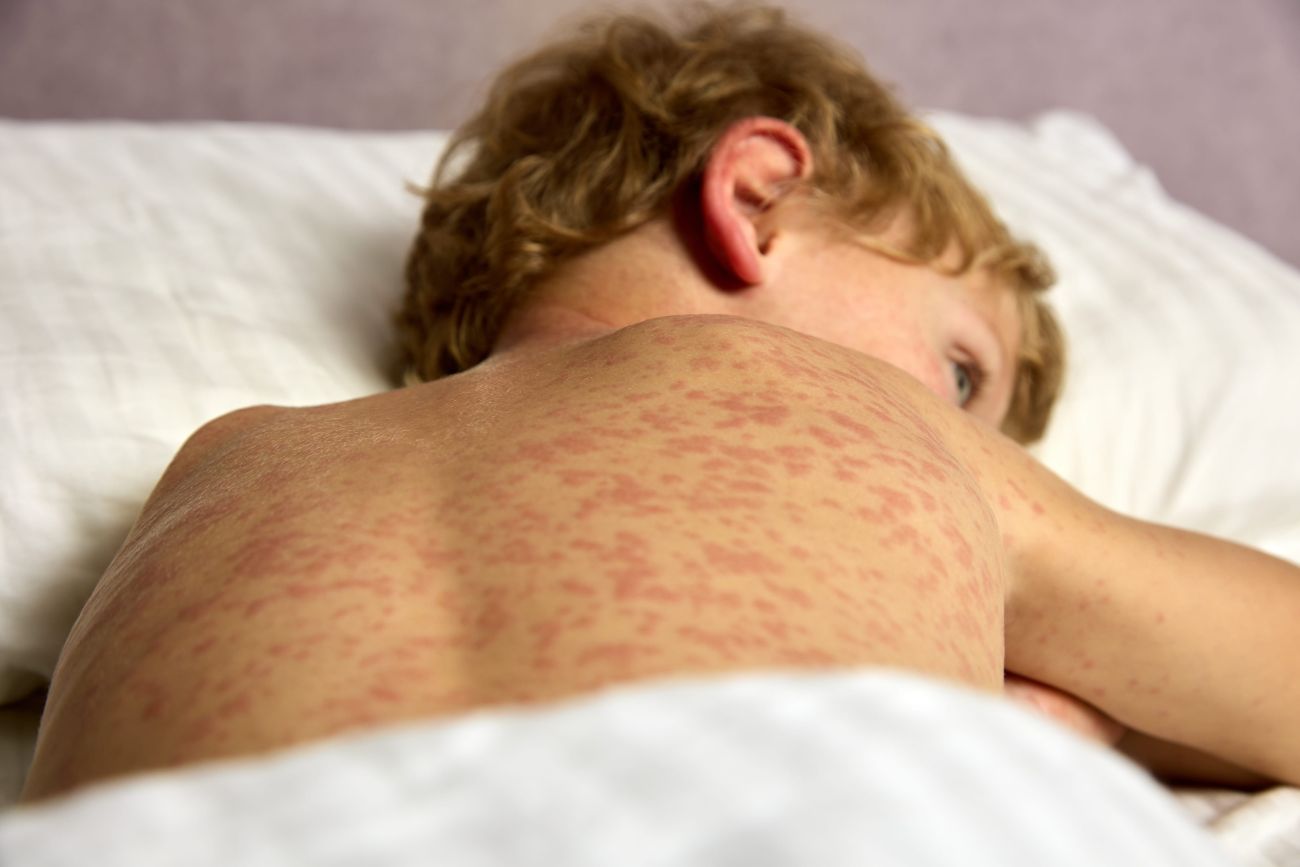
Prominent purpuric rash on the buttocks and legs in a child with Henoch-Schönlein purpura (IgA vasculitis). Such non-blanching, raised purpura in gravity-dependent areas is the hallmark of HSP.
IgA vasculitis (HSP) is the most common childhood vasculitis. It is a small-vessel vasculitis caused by IgA immune complex deposition, often triggered by an upper respiratory infection (commonly viral or streptococcal). Typical presentations include a triad of: (1) palpable purpura (small bruising or petechial rash) concentrated on dependent areas (usually legs and buttocks in ambulatory children) without thrombocytopenia, (2) arthritis/arthralgia (especially of the ankles or knees), and (3) abdominal pain. The rash is classically symmetric and palpable, often preceded by urticarial or erythematous lesions, and may be accompanied by edema (e.g., periorbital or peripheral edema in hands/feet, scrotal swelling in boys). Joint involvement occurs in about 50–75% of cases (usually transient pain or swelling in lower limb joints). Gastrointestinal involvement (seen in ~50%) can cause diffuse abdominal pain, GI bleeding, or even intussusception – intussusception is the most common GI complication of HSP. Renal involvement occurs in up to 25–50% of children, typically manifesting as hematuria and/or proteinuria due to IgA nephritis. Serious renal complications (nephritic or nephrotic syndrome, hypertension, renal failure) are less common but are the most concerning sequelae of HSP. Notably, kidney disease in HSP usually appears late (often 1–2 weeks after onset, rarely after 6 months) rather than at initial presentation.
Diagnosis: HSP is a clinical diagnosis based on the characteristic purpura and supportive features. Laboratory tests mainly help exclude other diagnoses and assess complications. Key initial tests include a complete blood count (CBC) to ensure normal platelet count—purpura with normal platelets favors HSP over thrombocytopenic purpura—and a urinalysis with blood pressure measurement to detect hematuria, proteinuria, or hypertension from renal involvement. In classic cases, these may be the only tests needed. If the presentation is atypical or severe, additional labs may be performed, such as inflammatory markers (ESR, CRP), renal function tests (BUN, creatinine, electrolytes), coagulation studies if bleeding is a concern, and titers for triggers like streptococcal infection (ASO). Autoimmune tests (ANA, dsDNA, ANCA, complement levels) are not routinely necessary unless there is significant renal involvement or an unclear diagnosis that suggests a different vasculitis. In HSP, platelet count and coagulation are generally normal, and IgA levels may be elevated (supportive but not diagnostic). As a biopsy is typically rarely required, it would show leukocytoclastic vasculitis with IgA deposition.
Management: HSP is typically self-limited and resolves within around 4 weeks in most cases. Therefore, management is primarily supportive for mild cases. Key aspects of treatment depend on severity:
- Mild cases (rash with minimal pain and no organ threat): Outpatient management with supportive care is sufficient. Ensure rest and hydration. Provide analgesics for pain: acetaminophen (paracetamol) or NSAIDs for arthralgias, if there are no contraindications. (NSAIDs should be avoided if there is significant GI bleeding or renal impairment.) Edema can be alleviated by leg elevation and bed rest.
- Moderate pain or more extensive symptoms (e.g., significant joint pain, severe abdominal pain, or early kidney involvement): Corticosteroids are often indicated to relieve symptoms. A typical regimen involves oral prednisolone at a dose of approximately 1–2 mg/kg/day (up to 60 mg/day) for a couple of weeks, followed by a taper once symptoms improve. Steroids can dramatically reduce the duration and intensity of abdominal pain and may alleviate severe joint pain or scrotal swelling. Important: corticosteroids do not prevent HSP nephritis or other long-term renal complications*. They are used for symptom control and to treat existing severe organ involvement, but they have not been shown to prophylactically reduce the incidence of kidney disease in HSP. Consider consulting a pediatric specialist when starting steroids or if moderate severity features are present.
- Severe disease (e.g., inability to maintain hydration, gastrointestinal complications such as intussusception or gastrointestinal hemorrhage, significant renal involvement, or other organ threats like pulmonary or neurological involvement): This necessitates hospitalization and specialist management. High-dose steroids (e.g., IV methylprednisolone) may be administered acutely. In cases of progressive kidney disease (nephritic/nephrotic syndrome) or other life-threatening features, aggressive immunosuppressive therapy is considered—such as IV pulse steroids combined with cyclophosphamide or azathioprine, IV immunoglobulin, or plasmapheresis, guided by a pediatric nephrologist or rheumatologist. Such interventions are beyond the scope of primary care and require referral.
Follow-up protocol: Close follow-up of HSP patients is essential to detect renal involvement that may arise after the acute symptoms resolve. Kidney disease in HSP can be insidious and asymptomatic in its early stages. A scheduled urinalysis and blood pressure monitoring plan should be implemented for at least 6 months after onset, as 90% of HSP nephritis cases begin within 2 months and 97% by 6 months. A practical follow-up schedule includes weekly checks during the first month, every 2 weeks during months 2–3, and then at 6 months and 12 months. At each visit, perform a dipstick urinalysis and measure blood pressure. If any hematuria, proteinuria, or hypertension is detected, or if the HSP rash or arthritis recurs, increase the monitoring frequency and seek further evaluation (including quantitative urine protein, renal function tests, and specialist referral). If the urinalysis remains normal (or shows only transient microscopic hematuria) throughout 6–12 months, long-term renal sequelae are very unlikely, and follow-up can be concluded. Parents should be informed that HSP can recur, with about 25–35% of children experiencing a recurrence within 4-6 months of the initial episode. Recurrences are typically milder and shorter, but they still warrant the same vigilance for renal involvement. GPs should arrange a prompt nephrology consultation if any red flags of renal disease emerge during follow-up (such as sustained proteinuria, rising creatinine, hypertension, or nephrotic/nephritic syndrome).
Prognosis: Most children with HSP recover fully with no lasting issues, especially if their kidneys are not affected. The rash and joint pain resolve within weeks, while abdominal pain typically eases in a few days once inflammation subsides. Even when the kidneys are involved, most show mild changes that remit over time. However, a small subset (1–5%) can progress to end-stage renal disease. Thus, the overall prognosis is excellent for most, but identifying the minority who develop serious nephritis is a key role of follow-up.
Kawasaki Disease
Kawasaki disease (KD) is an acute systemic vasculitis that affects medium-sized arteries, primarily impacting young children, particularly those aged 1 to 5 years, and leading to inflammation of the coronary arteries. KD ranks as the second most common pediatric vasculitis following HSP. Prompt recognition is essential, as untreated Kawasaki disease can result in coronary artery aneurysms in about 25% of cases, posing a risk of myocardial infarction or ischemia in young patients. However, with timely treatment, the risk of aneurysm decreases significantly to about 4% or lower. Kawasaki disease also stands as a leading cause of pediatric acquired heart disease worldwide.
Typical presentation: The hallmark is a fever lasting at least 5 days (often high spiking fevers of ≥39°C that do not respond to antipyretics), accompanied by a variety of mucocutaneous symptoms. The diagnostic criteria include a fever of ≥5 days plus at least 4 of the following 5 features:
- Bilateral conjunctival injection: Non-purulent conjunctivitis (red eyes without discharge), typically bilateral and sparing the limbus (white ring around the iris).
- Oropharyngeal changes: Mucosal inflammation such as “strawberry tongue” (red tongue with prominent papillae), red cracked lips, and diffuse oropharyngeal redness (without ulcerations or exudates).
- Polymorphous rash: A generalized rash that can be maculopapular, scarlatiniform, or erythema multiforme-like. It often starts on the trunk or diaper area and spreads; non-blanching (purpuric) or vesicular rashes are not typical of Kawasaki and suggest alternative diagnoses.
- Extremity changes: Redness and swelling of the hands and feet, often with painful edema of the palms and soles in the acute phase, followed by characteristic periungual desquamation (peeling of skin around fingertips and toes) in the subacute phase (usually in weeks 2–3). Infants might refuse to crawl or bear weight due to extremity pain.
- Cervical lymphadenopathy: Enlarged lymph node(s) in the neck, often unilateral, with at least one node >1.5 cm. This is the least consistently present feature (seen in fewer than half of cases, especially older children).
It’s important to note that not all children with KD meet the complete criteria. Symptoms can appear sequentially, and some may be subtle or transient. Incomplete Kawasaki disease is common in infants and adolescents, who may experience prolonged fever with only 2–3 of the criteria. These patients are at a higher risk for coronary complications than those with full (classic) presentations. Thus, a high index of suspicion is necessary; clinicians should consider KD in any child with unexplained persistent fever (especially if it lasts more than 5 days) and any of the above features, even if not all criteria are satisfied. Infants under 6 months with fever lasting 7 days or more and no clear source should raise concern for Kawasaki disease, even if other signs are absent. Extreme irritability in a febrile child is another clue; KD patients are often notably irritable (more so than in typical viral illnesses).
Complications: The most serious complication is coronary arteritis, which can lead to aneurysm formation. Coronary dilation might begin by the 10th day of illness, and if untreated, about 1 in 4 children may develop coronary aneurysms (with ~1% developing giant aneurysms >8 mm, which carry the highest risk of thrombosis). Aneurysms can occasionally occur in other arteries (e.g., axillary) as well. Kawasaki disease can also cause myocarditis, arrhythmias, heart failure, valvular regurgitation, or, rarely, myocardial infarction and sudden death in the acute phase. These complications typically occur in the subacute phase (2–4 weeks in), when coronary inflammation alongside a hypercoagulable state peaks. The case-fatality rate of KD is low (~0.17% in-hospital), but virtually all deaths result from cardiac complications. This is why early treatment is vital.
Initial evaluation and labs: There is no specific diagnostic test for Kawasaki disease; diagnosis is made clinically. However, certain lab findings are characteristic and can support the diagnosis, especially in incomplete cases. These include elevated acute inflammatory markers (high ESR and CRP), neutrophil-predominant leukocytosis, normocytic anemia, hypoalbuminemia, and mildly elevated liver enzymes (often ALT). Platelet count is typically normal in the first week but then rises; thrombocytosis (often very high platelet count) is a hallmark in the second week of illness. (This late thrombocytosis is not helpful early on, but a normal-to-low platelet count in a child beyond day ~7 of fever makes KD less likely.) Urinalysis may show sterile pyuria (white cells in urine without bacterial growth). If Kawasaki disease is suspected, GPs should not delay referral while awaiting lab results; labs can assist in ambiguous cases, but clinical judgment and early treatment are paramount.
Management: Urgent referral and inpatient treatment is necessary whenever Kawasaki disease is suspected. As a GP, do not “wait and see” beyond 5 days of fever if KD is in the differential – early intervention is crucial. The standard first-line treatment is intravenous immunoglobulin, administered at a dose of 2 g/kg given as a single infusion, ideally within 10 days of fever onset. IVIG dramatically reduces the risk of coronary aneurysms from approximately 25% to about 2–5%. Even if a child presents after day 10, IVIG should still be administered if there is ongoing fever or inflammation, as it can still provide benefits. IVIG typically causes rapid defervescence (fever resolution) and improvement in clinical symptoms for the majority of cases. Alongside IVIG, high-dose aspirin therapy is traditionally used during the acute phase of involvement. A typical regimen involves high-dose aspirin (approximately 30–50 mg/kg/day, divided, or approximately 80–100 mg/kg daily), followed by a transition to a low-dose regimen, such as platelet-thrombin inhibitors (3–5 g/kg daily), once the fever subsides. The role of high-dose aspirin is anti-inflammatory (though evidence is not from RCTs), while the low-dose phase aims to prevent thrombosis until the coronary risk has passed. If the child shows no coronary abnormalities on the follow-up echocardiogram at 6–8 weeks, specialists can generally discontinue aspirin at that point. (If d, longer-term aspirin or additional anticoagulation will be managed by cardiology.)
Refractory cases (persistent fever after initial IVIG) may receive a second IVIG infusion. If IVIG fails, corticosteroids (e.g., IV methylprednisolone pulses) or other immunomodulators, such as tumor necrosis factor (TNF) inhibitors, can be used as second-line treatments. These decisions are made in the hospital by specialists. GPs should be aware of these possibilities; however, the key role of GPs is timely recognition and referral, rather than administering these therapies.
Follow-up and GP role after acute treatment: All Kawasaki disease patients require cardiology follow-up with echocardiography to monitor their coronary arteries. Typically, an echo is performed at diagnosis, around 6–8 weeks after the onset of the illness, and periodically thereafter depending on the degree of coronary involvement. For a child who had normal or only transient coronary changes, the 6–8 week echo may be the last one; whereas those with aneurysms need serial echoes and long-term cardiology care. The GP should ensure that these cardiology appointments are arranged and that the family understands their importance.
After being discharged from the hospital, the child is often sent home on a low dose of aspirin until the cardiology team advises discontinuation (typically at the 6–8 week follow-up if no aneurysms are present). The GP should counsel parents about aspirin safety: watch for signs of bleeding, and be aware of the very small risk of Reye syndrome if the child contracts influenza or varicella while on aspirin. Because of this, annual influenza and varicella vaccinations are recommended for children on long-term aspirin if they are not already immune. Additionally, IVIG can interfere with live vaccines; specifically, measles and varicella vaccines must be deferred for about 11 months after IVIG administration because the passive antibodies can blunt the vaccine response. The GP should coordinate with immunization schedules: for example, if the child is due for an MMR or varicella vaccine within that 11-month window, it should be postponed. Non-live vaccines, like influenza, can be given as scheduled. These nuances should be communicated to the family at discharge; however, reinforcing them during GP follow-up visits is valuable.
GPs can also reassure parents that Kawasaki disease is not contagious, and with appropriate treatment, the vast majority of children recover without long-term problems. About 50% of coronary aneurysms (if they occur) will resolve within 1–2 years, though some arterial damage may persist. Long-term, if a child has significant coronary involvement, they will need ongoing cardiology oversight (and management of cardiovascular risk factors as they grow). Fortunately, true recurrence of Kawasaki disease is rare (it can recur in a small percentage of patients, but most children do not get it again). Any child with a history of KD who presents with new febrile illness and skin changes should be evaluated carefully, but benign peeling of skin with subsequent fevers can occur and doesn’t necessarily indicate KD recurrence.
Bottom line for GPs: If you encounter a child with an unexplained fever lasting 5 days or more, especially if accompanied by a rash, red eyes, red lips or tongue, swollen extremities, or extreme irritability, consider Kawasaki disease. This constitutes an emergency – arrange for prompt pediatric assessment. Early IVIG treatment within 10 days is the proven measure that significantly reduces the risk of coronary artery aneurysms. Do not delay referral while waiting for all criteria to be met or for lab results if clinical suspicion is high. It is better to refer and have Kawasaki disease ruled out than to miss it and risk cardiac complications.
Other Pediatric Vasculitides (Brief Overview)
Beyond HSP and Kawasaki disease, other vasculitides in children are rare but significant. GPs should recognize their existence, as initial signs may be subtle, and these conditions typically necessitate specialist referral and management. A few examples include:
- ANCA-Associated Vasculitis: This category includes Granulomatosis with Polyangiitis (GPA, formerly Wegener’s) and Microscopic Polyangiitis (MPA). These are small-vessel vasculitides that are quite rare in young children (more often seen in adolescents). They may present with unexplained chronic ENT symptoms (sinusitis, otitis), lung involvement (persistent cough, pulmonary infiltrates, or hemorrhage), renal glomerulonephritis (hematuria, proteinuria, renal failure), and systemic symptoms (fever, weight loss). The presence of both respiratory and kidney findings should raise concern for ANCA-associated vasculitis. c-ANCA (PR3) is often positive in GPA. Any child suspected of GPA/MPA requires prompt referral to pediatric rheumatology/nephrology; treatment involves aggressive immunosuppression (steroids, cyclophosphamide, or rituximab, etc.).
- Polyarteritis Nodosa (PAN) is a medium-vessel necrotizing vasculitis that is very rare in children. It can present with a wide range of findings, including systemic illness (fever, weight loss, malaise), cutaneous lesions (nodules, livedo reticularis, purpura), renal involvement (hypertension due to renal artery microaneurysms), abdominal pain (mesenteric ischemia or microaneurysms), myalgias/arthritis, and, in some cases, testicular pain in boys (due to vasculitis of testicular arteries). In some cases, PAN has been associated with hepatitis B infection. Any child suspected of having PAN should be referred for confirmatory testing and management, which often requires angiography for diagnosis and immunosuppressive therapy.
- Takayasu Arteritis is a large-vessel granulomatous vasculitis that primarily affects the aorta and its branches. It is exceedingly rare in the pediatric age group, typically occurring in adolescent girls. Clues include unexplained hypertension, discrepancies in blood pressure or pulses between arms, bruits over the subclavian arteries or aorta, and systemic symptoms such as fever, fatigue, and weight loss. Takayasu arteritis can initially present with nonspecific signs, so high suspicion is necessary if a teenager shows unusual blood pressure findings or experiences limb claudication. A referral for vascular imaging (MRI/CT angiography) is required if it is suspected.
- Behçet Disease: A rare variable-vessel vasculitis that primarily affects adolescents and specific ethnic groups. It is characterized by recurrent painful oral and genital ulcers, eye inflammation (uveitis), and may present with skin lesions (such as erythema nodosum), arthritis, or even neurological symptoms. Any child exhibiting the classic triad of oral ulcers, genital ulcers, and eye issues should be referred to a specialist, as Behçet necessitates immunomodulatory therapy.
- Others: There are even rarer pediatric vasculitides (e.g., primary angiitis of the central nervous system presenting with neurologic deficits, cryoglobulinemic or hypersensitivity vasculitis presenting with rash and possibly kidney issues, etc.). These conditions are outside the scope of primary care for diagnosis, but general practitioners should consider vasculitis in the differential diagnosis when a child presents with unexplained systemic illness involving multiple organs.
In summary, any suspected vasculitis outside of the straightforward HSP realm requires specialist evaluation. GPs should concentrate on identifying red flag features and initiating the referral process. We will now highlight general red flags and approach.
Red Flags: When to Consider Vasculitis in a Child
General practitioners should consider vasculitis in a pediatric patient when observing the following red flags or clinical scenarios:
- Palpable purpura or unexplained petechial rash with normal platelets: Non-blanching purpura in a well-appearing child suggests a small-vessel vasculitis like HSP, rather than low platelets (ITP) or meningococcemia. Any child with unexplained purpura (especially if localized to gravity-dependent areas and not associated with thrombocytopenia) should be evaluated for HSP.
- Prolonged fever with no clear source: A fever lasting five days or more, particularly if unresponsive to antibiotics and antipyretics, is a warning sign. If such a fever is accompanied by any mucocutaneous findings (rash, conjunctivitis, red tongue/lips, swollen hands/feet), Kawasaki disease must be high on the differential list. GPs should also consider incomplete KD in infants with seven or more days of fever even if classic signs are absent.
- Extreme irritability in a febrile child: Disproportionate irritability can indicate Kawasaki disease—parents may report the child is “inconsolable” or unusually fussy. This should prompt a careful examination for other KD features.
- Multisystem involvement: Any combination of symptoms affecting multiple organ systems without another unifying diagnosis should raise the question of vasculitis. For example, skin lesions plus renal findings (e.g., purpura with hematuria/proteinuria suggests HSP or ANCA vasculitis); lung symptoms plus renal failure (consider ANCA-associated vasculitis); unusual neurological symptoms (e.g., strokes) plus systemic inflammation (could be primary CNS vasculitis or others); limb claudication or absent pulses (consider Takayasu).
- Unusual or severe manifestations of common symptoms: For instance, unexplained testicular pain (HSP or PAN can cause orchitis/infarction), digital ischemia or gangrene (could be medium-vessel vasculitis), or skin ulcers/nodules with systemic signs (could indicate PAN or ANCA vasculitis).
- Recurrent fevers with rash or oral ulcers: Recurrent patterns (especially with mucosal ulcers, as in Behçet) may indicate an autoinflammatory or vasculitic process rather than simple viral infections.
In practice, a good rule of thumb is: if a child presents with an unexplained constellation of findings (fever, rash, unusual pain, lab abnormalities) that don’t align with a single common illness, consider vasculitis and order basic screening tests.
Initial Workup in Prihelp confirms litis is part of the statistical diagnosis; a targeted initial workup can assist in establishing the diagnosis and evaluating the extent of the disease. GPs should conduct the following common investigations while simultaneously arranging a referral, if needed:
- Vital signs, particularly blood pressure: Hypertension in a child with suspected vasculitis (especially HSP or PAN) is a concerning indicator of renal or vascular involvement and must be documented and addressed.
- Urinalysis: This is a crucial test for any child with possible vasculitis, notably HSP or ANCA-associated vasculitis. Check for hematuria and proteinuria. Even if a child with HSP appears well, a dipstick urinalysis can uncover renal involvement that might otherwise be overlooked (as early nephritis is often asymptomatic). The presence of RBCs or protein requires further quantification (protein/creatinine ratio) and possibly blood tests for kidney function.
- Complete Blood Count (CBC) with platelets: Assess platelet count (normal or high in vasculitis vs. low in ITP), white blood cell count (often elevated in systemic vasculitis or Kawasaki, with neutrophil predominance), and hemoglobin (anemia of chronic disease is common in prolonged inflammation; also, severe anemia could indicate bleeding in HSP gastrointestinal involvement).
- Acute phase reactants: ESR and CRP are typically elevated in active vasculitis. A high CRP/ESR in a febrile child with a rash strengthens the case for an inflammatory or vasculitic process (for example, high ESR/CRP is expected in Kawasaki, while in a simple viral exanthem, they would be only mildly elevated, if at all).
- Metabolic panel: If renal involvement is suspected, check BUN, creatinine, and electrolytes. In HSP with significant proteinuria or in ANCA vasculitis, signs of renal impairment may be evident. Liver enzymes can be assessed if Kawasaki is a possibility (mild ALT elevation is common).
- Serologies and specific tests: These are generally guided by the clinical context. For HSP, you might check an ASO titer or anti-DNase B to determine if a recent strep infection could have triggered the vasculitis. For suspected Kawasaki, there is no specific serology; however, COVID-19 antibody testing might be considered in the 2020+ era if MIS-C is suspected (which can mimic KD). For suspected ANCA-associated vasculitis, send ANCA titers (c-ANCA/PR3 for GPA, p-ANCA/MPO for MPA). ANA/dsDNA might be tested if considering lupus vasculitis versus HSP in a teenager with renal issues. Complement levels (C3, C4) may be low in lupus or certain cryoglobulinemic vasculitis but normal in HSP. These specialized tests are typically not first-line in primary care but can be performed if the referral wait is long and clinical suspicion is high.
- Imaging when appropriate: If a child with HSP experiences severe abdominal pain, an abdominal ultrasound should be performed to check for intussusception or bowel ischemia. If Kawasaki disease is suspected and the child is in an outpatient setting, arranging an echocardiogram immediately is not necessary before referral (the hospital will conduct it); however, if there’s a delay in specialty care, an echo can be beneficial. For suspicion of Takayasu arteritis, vascular imaging (MRI/CT angiography) would be arranged by specialists, not initially by the general practitioner.
Importantly, these investigations should not delay referral in an urgent case. For instance, if Kawasaki disease is likely, perform basic labs if quickly available, but do not wait for results—get the child to a facility where IVIG can be administered. Likewise, if a child is very ill or you suspect severe vasculitis (renal failure, pulmonary hemorrhage, etc.), arrange for emergent transfer.
When to Refer to a Specialist or Hospital
Because pediatric vasculitis can involve critical organs, the threshold for involving specialists or hospitalizing the patient is low. Here are some general guidelines on when GPs should refer:
- Any suspicion of Kawasaki disease warrants an URGENT referral. Kawasaki should be managed in a hospital by experienced clinicians. If a child has a fever lasting more than 5 days with features suggestive of KD (even if incomplete), refer them to pediatric emergency or cardiology/rheumatology without delay. Guidelines recommend referral for any child with prolonged fever and clinical features without alternative explanation, or any infant under 6 months with a fever lasting more than 7 days (possible incomplete KD). Early referral is crucial because IVIG should ideally be started between days 5 and 7 (certainly by day 10) of the illness.
- For Henoch-Schönlein purpura, refer or admit if significant complications or certain risk factors are present. Indications for hospitalization or consultation include severe abdominal pain or gastrointestinal bleeding (risk of intussusception or ischemia), inability to maintain hydration, severe joint pain that prevents ambulation, any neurologic symptoms (which could indicate rare cerebral vasculitis or hemorrhage), any pulmonary involvement (e.g., hemoptysis), and severe renal involvement such as macroscopic hematuria, nephrotic-range proteinuria, rising creatinine, or hypertension. Essentially, mild HSP can be managed as an outpatient, but if the child has more than mild symptoms or any organ-threatening features, involve a pediatric team. Starting corticosteroids in HSP is also a consideration for inpatient care or at least specialist input. If urinalysis reveals more than a trace of blood (e.g., gross hematuria or significant proteinuria), the child should be co-managed with or evaluated by a pediatric nephrologist. Persistent proteinuria or hypertension requires nephrology consultation.
- ANCA-associated vasculitis or systemic polyarteritis nodosa must always be referred to specialists. If a GP suspects ANCA vasculitis (for example, due to lung and kidney involvement with +ANCA), this is a medical emergency often requiring hospitalization for immunosuppressive therapy. If polyarteritis nodosa is suspected (e.g., a child with systemic illness, hypertension, and skin nodules), it should prompt an urgent referral to pediatric rheumatology; often, these patients will need a workup for organ ischemia (e.g., angiography) and treatment with high-dose steroids and cyclophosphamide.
- Any evidence of significant organ ischemia or damage in a child—e.g., digital gangrene, serious neurologic deficit (stroke/seizure of unclear cause in systemic inflammation)—should raise suspicion for vasculitis and warrants emergent referral.
- Uncertain diagnosis but red flags present: If you’re unsure what is happening, but the child shows concerning signs (fever, rash, lab abnormalities) that could indicate vasculitis, it is wise to refer to a tertiary center or pediatric rheumatologist for further evaluation. It is better to err on the side of referral in pediatrics, considering the potential for rapid deterioration.
Remember that as a GP, you don’t need to definitively diagnose which vasculitis before referring; if the signs indicate a vasculitic process beyond a mild self-limiting case, involve specialists early. Parents should be advised to seek immediate care (or the GP should arrange direct admission) if the child develops any new alarming symptoms such as severe abdominal pain (which could indicate intussusception in HSP), chest pain or breathing difficulties (which could indicate pulmonary hemorrhage or coronary issues in KD), severe headaches or neurological changes (CNS vasculitis or hemorrhage), or gross blood in the urine. All these warrant urgent evaluation.
Management and Follow-Up: GP’s Role
The role of the GP in pediatric vasculitis includes early identification, initial management of mild cases, coordination of specialist care, and long-term follow-up once the acute phase has been addressed.
1. Initial management in primary care: For mild presentations (primarily HSP), GPs can manage symptoms and closely observe patients. Ensure adequate pain control (paracetamol/NSAIDs for HSP aches if no contraindications). Encourage hydration and rest. If you decide to start a course of steroids for HSP due to moderate symptoms, do so in consultation with a pediatrician when possible, and have a follow-up plan. For any more serious presentations, initiate stabilization (e.g., pain medication, blood pressure control if severe hypertension, IV fluids if dehydrated) while arranging transfer. GPs may also initiate empirical antibiotics if bacterial infection is a consideration (e.g., some Kawasaki Disease cases receive antibiotics initially for possible adenitis or scarlet fever until the diagnosis is confirmed; however, once Kawasaki Disease is recognized, antibiotics should be discontinued). Always address any emergency issues first (for example, a child with HSP and signs of intussusception needs immediate surgical consultation).
2. Education and support: Explain the condition to the family in understandable terms. For HSP, parents should understand that the disease is usually benign and resolves, but they must adhere to follow-up to check the kidneys. Provide guidance on signs of complications (e.g., worsening abdominal pain, blood in urine) that should prompt immediate return. For Kawasaki Disease, families should understand the need for hospital treatment and the importance of follow-up cardiology visits. They should be advised to avoid giving NSAIDs like ibuprofen without checking with doctors if the child is on aspirin therapy, and to inform healthcare providers in the future that the child had Kawasaki Disease (as certain precautions with procedures or medications might apply).
3. Outpatient follow-up and long-term monitoring: Following the acute illness, general practitioners (GPs) play a crucial role in continued monitoring.
- For HSP, as detailed earlier, set up a schedule of blood pressure checks and urinalysis for over six months. This can often be performed at the primary care clinic. If anything abnormal is found (e.g., 2+ protein or blood on dipstick, or blood pressure above the normal range for age and height), repeat the test and consider sending the results to nephrology for further evaluation. If all follow-up checks are normal through six to twelve months, you may discharge the patient from further surveillance.
- For Kawasaki Disease, ensure the child attends cardiology follow-ups (typically an echocardiogram at six to eight weeks post-onset, and possibly another at one year if any concerns arise). If the child is on low-dose aspirin at home, the GP should monitor for any side effects (easy bruising, including bleeding, gastrointestinal irritation) and ensure appropriate vaccination monitoring and scheduled (annual flu if not immune). Reminding parents about tracking delays in live vaccines post-IVIG is essential. After the six-week cardiology visit, the cardiologist will advise whether aspirin can be ceased or needs to continue. If any cardiac issues were found (even transient coronary dilation), the child may need a longer follow-up; if none, they likely return to normal pediatric care. In either case, general pediatric care should include advising a heart-healthy lifestyle as the child grows (since severe Kawasaki Disease can have long-term implications for coronary health).
- For other vasculitides, follow-up is typically co-managed with specialists. For example, a child with ANCA vasculitis may be on immunosuppressants for a prolonged duration. The GP should help monitor blood counts, liver and kidney function according to the specialist’s plan, and watch for medication side effects or infections. Regular blood pressure monitoring is crucial for any child who had renal involvement. GPs also provide routine healthcare maintenance that chronic vasculitis patients need: ensuring catch-up on immunizations (with adjustments if on immunosuppressives), growth and development monitoring (high-dose steroids can affect growth, so track growth curves), and psychosocial support (chronic illness can be challenging for families).
4. Coordination of care: Act as a liaison between the family and specialists. Ensure that the family knows which symptoms merit returning sooner than scheduled. For instance, in a child with known vasculitis on therapy, a fever could indicate infection (due to immunosuppression) or disease flare – the GP should coordinate appropriate evaluation. Keep clear documentation of the vasculitis diagnosis in the child’s record for future reference, and communicate with the child’s other healthcare providers (e.g., if a surgeon or another specialist sees the patient, they should be aware of the vasculitis history and current medications).
5. “Red flag” vigilance during follow-up: Even after initial recovery, GPs should remain alert for late-emerging issues. For HSP, late renal involvement is the main concern (hence the six-month surveillance). For Kawasaki, late-developing aneurysms are rare but can occur (some coronary changes might only become evident after a couple of weeks, which is why an echocardiogram at week six to eight is performed). Any new symptoms in a child who has had vasculitis should be interpreted in light of that history – e.g., a new rash in a recent HSP patient could indicate a recurrence of HSP; chest pain in a recent Kawasaki patient should prompt an urgent cardiology evaluation, etc.
In conclusion, general practitioners play a crucial role in the care of children with vasculitis. By recognizing typical presentations (palpable purpura of HSP, prolonged fever of Kawasaki, etc.) and red flags, ordering initial labs (CBC, ESR, CRP, UA) to support their suspicions, and knowing when to refer for specialized care, GPs ensure timely diagnosis and treatment. They also provide continuity through follow-up, reinforcing treatment plans and monitoring for complications or recurrence. Most pediatric vasculitides, with the notable exception of severe cases, have good outcomes if managed properly. The GP’s awareness and diligence can prevent missed diagnoses (like “missed” Kawasaki disease leading to young adult coronary disease) and improve long-term health for these children.
Sources:
- Royal Children’s Hospital Melbourne – Henoch-Schönlein Purpura (IgA Vasculitis) Clinical Practice Guideline
- American Academy of Family Physicians – Saulsbury, F.T. Henoch-Schönlein Purpura in Children
- Royal Children’s Hospital Melbourne – Kawasaki Disease Clinical Practice Guideline
- Royal Australian College of GPs (RACGP) – Golshevsky et al. Kawasaki disease: prompt recognition and early referral
- RCH Melbourne – HSP Parent Information & Starship Children’s Health HSP resource (follow-up record)
- American Heart Association – Kawasaki Disease Treatment and Outcomes (as cited in RACGP article)
- Additional references: Mayo Clinic and Vasculitis Foundation resources for general vasculitis knowledge; UpToDate – Vasculitis in Children: An Overview. (These provide general epidemiology confirming HSP and KD as the most common pediatric vasculitides.)
Top 10 Key Publications in Pediatric Vasculitis (2015–2025)
- European consensus-based recommendations for diagnosis and treatment of immunoglobulin A vasculitis – the SHARE initiative. Seza Ozen et al. (2019). Rheumatology (Oxford). – A pan-European expert panel issued seven diagnostic and 19 therapeutic recommendations for IgA vasculitis (Henoch-Schönlein purpura) in children. Key points include the appropriate use of skin and renal biopsies and imaging for diagnosis, supportive care for mild cases, and judicious use of corticosteroids for severe gastrointestinal or renal involvement. These guidelines aim to standardize and improve care for pediatric IgA vasculitis across centers. DOI: 10.1093/rheumatology/kez041
- Diagnosis, Treatment, and Long-Term Management of Kawasaki Disease: A Scientific Statement for Health Professionals from the American Heart Association. Brian W. McCrindle et al. (2017). Circulation. – This comprehensive guideline updates the 2004 AHA recommendations on Kawasaki disease, which is the most common cause of acquired heart disease in children. It provides an algorithm to help recognize incomplete Kawasaki presentations and emphasizes prompt IVIG therapy as the initial treatment. The statement also discusses adjunctive therapies (e.g., corticosteroids in high-risk cases) for primary treatment and the management of IVIG-resistant cases. Long-term management is stratified by coronary aneurysm z-scores, with lifelong cardiology follow-up for those with aneurysms. DOI: 10.1161/CIR.0000000000000484
- European consensus-based recommendations for the diagnosis and treatment of rare paediatric vasculitides – the SHARE initiative. Nienke de Graeff et al. (2019). Rheumatology (Oxford). – This guideline addresses less common pediatric vasculitides, including childhood polyarteritis nodosa (PAN), Takayasu arteritis, and ANCA-associated vasculitides (Wegener’s granulomatosis/GPA, microscopic polyangiitis, and eosinophilic granulomatosis). A total of 78 consensus recommendations were developed, covering disease-specific diagnostic workups and treatments. Notably, the guidance endorses the early use of immunosuppressants (e.g., cyclophosphamide, methotrexate, biologics) alongside steroids for severe cases, and emphasizes the importance of cross-disciplinary care (e.g., imaging for large-vessel vasculitis). These recommendations fill a significant gap, aiming for consistent, high-quality care in these rare pediatric conditions. DOI: 10.1093/rheumatology/key322
- Phase IIa global study evaluating rituximab for the treatment of pediatric patients with granulomatosis with polyangiitis or microscopic polyangiitis. Paul Brogan et al. (2022). Arthritis & Rheumatology. – This international open-label trial (PePRS study) demonstrated that rituximab is effective and well-tolerated in children with ANCA-associated vasculitis (GPA/MPA). By 12 months, 92% of pediatric patients had achieved disease remission (as measured by the Pediatric Vasculitis Activity Score), increasing to 100% by 18 months. No new safety signals were observed in children compared to adults; predominantly, adverse events were mostly mild, including infusion-related reactions. These results support the use of rituximab as a remission-induction and maintenance therapy in pediatric ANCA vasculitis, offering a steroid-sparing alternative to cyclophosphamide. DOI: 10.1002/art.41901
- Early-onset stroke and vasculopathy are associated with mutations in ADA2. Qing Zhou et al. (2014). New England Journal of Medicine. – This landmark study identified recessive mutations in the ADA2 gene (CECR1) as the cause of a novel monogenic vasculitis syndrome, now recognized as Deficiency of Adenosine Deaminase 2 (DADA2). Affected children presented with systemic inflammation, recurrent ischemic strokes, livedoid rash, and features resembling polyarteritis nodosa. The authors demonstrated that ADA2 mutations lead to markedly reduced ADA2 enzyme activity and a vasculopathy characterized by endothelial damage and inflammation. This discovery has a direct clinical impact: children diagnosed with PAN are now screened for DADA2, and those affected respond well to anti-TNF biologic therapy instead of traditional cytotoxic agents. DOI: 10.1056/NEJMoa1307361
- Infliximab versus second intravenous immunoglobulin for treatment of resistant Kawasaki disease in the USA (KIDCARE): a randomised, multicentre trial. Jane C. Burns et al. (2021). Lancet Child & Adolescent Health, with. – This multi-center RCT compared a single administration of infliximab to a second IVIG in children with Kawasaki disease, and in IVIG-treated children with Kawasaki disease. Infliximab significantly improved early outcomes: 77% of children in the infliximab group achieved fever resolution within 24 hours (with no fever recurrence) versus 51% in the second IVIG group. The infliximab-treated group also demonstrated trends toward shorter hospital stays and faster normalization of inflammation. Importantly, no increase in adverse events was observed with infliximab, supporting its use as a safe and effective escalation therapy for refractory Kawasaki disease. DOI: 10.1016/S2352-4642(21)00270-4
- Interventions for preventing and treating kidney disease in IgA vasculitis (Henoch-Schönlein purpura). Deirdre Hahn et al. (2015, updated 2023). Cochrane Database of Systematic Reviews. – This rigorous systematic review assessed 20 trials involving 1,963 patients to determine whether any therapies improve renal outcomes in IgA vasculitis. For children with mild disease, prophylactic prednisone or antiplatelet agents did not significantly prevent kidney involvement. In established nephritis, no immunosuppressive regimen— including cyclophosphamide, mycophenolate, or others— proved clearly superior in inducing remission, although data were limited. An ACE inhibitor (fosinopril) was beneficial in reducing proteinuria. Overall, the review concluded that there is no solid evidence that early corticosteroids alter long-term nephritis risk, affirming that steroids should not be used routinely for prophylaxis in HSP. These findings support a conservative, supportive approach for most pediatric cases, reserving aggressive therapies for proven severe nephritis. DOI: 10.1002/14651858.CD005128.pub3
- 2021 American College of Rheumatology/Vasculitis Foundation Guideline for the Management of Polyarteritis Nodosa. Sharon A. Chung et al. (2021). Arthritis Care & Research. – This ACR-endorsed guideline offers evidence-based recommendations for systemic PAN (not due to hepatitis B). While acknowledging the lack of pediatric-specific trials, it advises using high-dose corticosteroids in combination with cyclophosphamide for the initial treatment of severe PAN to induce remission quickly. To limit toxicity, it recommends tapering and switching to steroid-sparing agents (e.g., methotrexate or azathioprine) for maintenance once the disease is controlled. The guidance also emphasizes the appropriate use of biopsy and vascular imaging to confirm diagnosis. Notably, screening for DADA2 (in pediatric PAN) is now part of the workup in practice, although the management of that genetic subset involves biologics (an area outside the scope of this guideline). DOI: 10.1002/acr.24633
- 2021 American College of Rheumatology/Vasculitis Foundation Guideline for the Management of Giant Cell Arteritis and Takayasu Arteritis. Mehrdad Maz et al. (2021). Arthritis & Rheumatology. – This guideline, although focusing primarily on large-vessel vasculitis in adults, incorporates insights from pediatric rheumatologists regarding Takayasu arteritis (a condition often presenting during adolescence). It strongly recommends the early use of nonglucocorticoid immunosuppressants (e.g., methotrexate or TNF inhibitors) in conjunction with high-dose glucocorticoids for Takayasu arteritis to enhance outcomes. Imaging techniques (MRI, CT, or PET angiography) are advised for the diagnosis and monitoring of large-vessel inflammation. Due to limited trial data, most recommendations are conditional, yet they establish a framework to balance aggressive disease management with minimizing cumulative steroid exposure in young patients. DOI: 10.1002/art.41774
- 2021 American College of Rheumatology/Vasculitis Foundation Guideline for the Management of ANCA-Associated Vasculitis. Sharon A. Chung et al. (2021). Arthritis Care & Research. – This is the first ACR guideline on ANCA-associated vasculitides and is relevant to older children and adolescents with GPA (Wegener’s), MPA, or EGPA. It recommends rituximab (along with glucocorticoids) as the first-line induction therapy for severe GPA/MPA, and also for maintenance of remission—reflecting evidence that RTX is as effective as cyclophosphamide with fewer long-term side effects. For eosinophilic GPA, add-on therapy with mepolizumab (an IL-5 inhibitor) is recommended in non-severe cases to assist in tapering steroids. All recommendations are conditional due to limited pediatric-specific data, but this guidance provides a practical, up-to-date approach that aligns pediatric practice with adult trial evidence. DOI: 10.1002/acr.24634